I often find myself gazing at the night sky, a vast canvas of familiar stars and galaxies, and wondering: what else is out there that we haven't even conceived of? Our understanding of the universe, profound as it is, is built on the elements we know—the tidy rows of the periodic table, painstakingly discovered and categorized here on Earth. But what if the cosmos, in its boundless and extreme environments, harbors elements that defy our current scientific paradigms? Could there be "impossible" elements, born in the hearts of dying stars or within the crushing gravity of neutron stars, waiting to be discovered?
This isn't a flight of pure fantasy. The notion of undiscovered elements pushes the boundaries of nuclear physics and astrophysics, leading us to contemplate how matter itself behaves under conditions utterly alien to our terrestrial labs. I'm talking about elements that might be incredibly unstable, existing for mere fractions of a second, or perhaps stable only under immense pressures and temperatures, hinting at a chemistry far more complex than we can currently imagine.
The Known Universe of Elements: A Quick Refresher
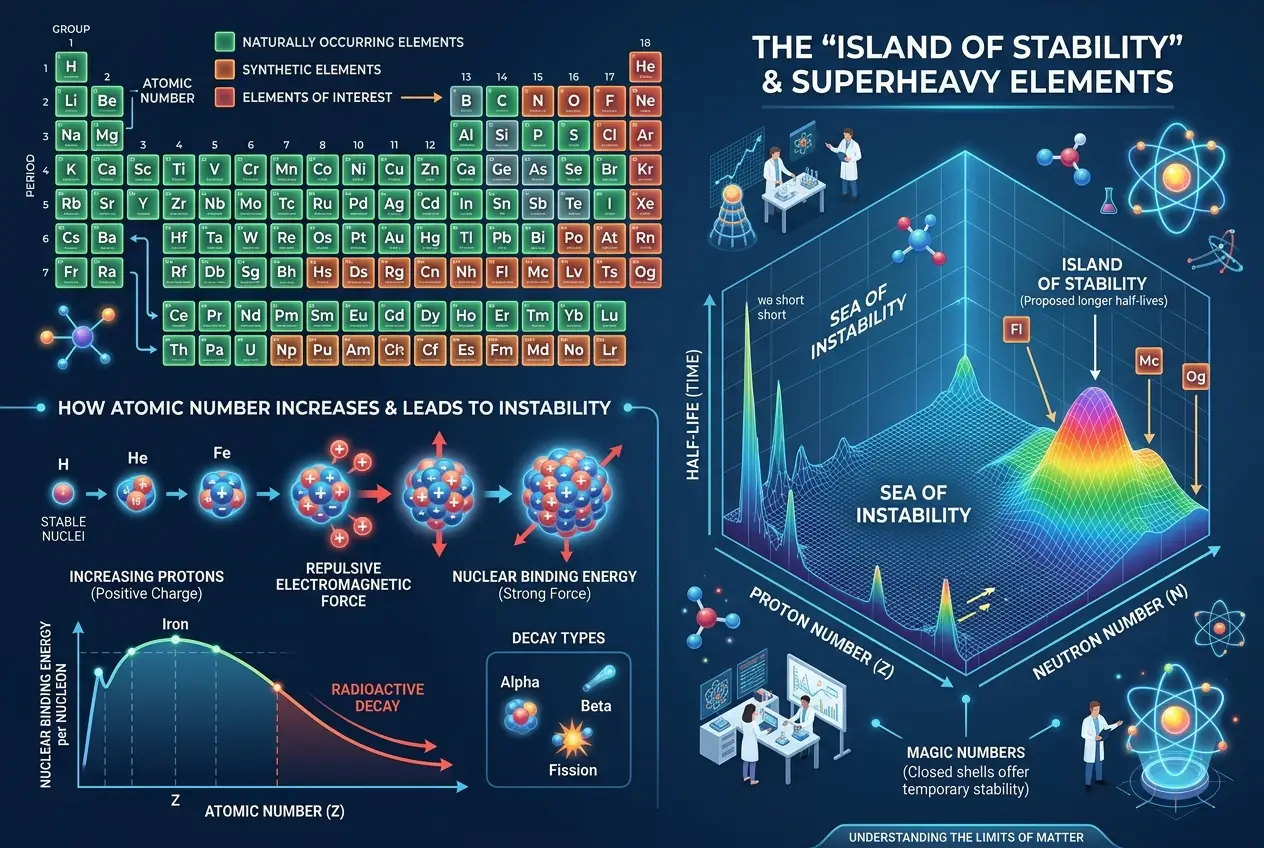
Before we venture into the truly impossible, let's briefly ground ourselves in what we *do* know. The periodic table, a triumph of human scientific endeavor, organizes elements by their atomic number—the number of protons in their nucleus. From hydrogen (1 proton) to Oganesson (118 protons), each element has a unique identity, defined by its electron configuration, which dictates its chemical properties.
Naturally occurring elements stop at uranium (92 protons). Beyond that, elements are synthetic, created in particle accelerators, often decaying almost instantly. These **transuranic elements** are fascinating, pushing the limits of nuclear stability. But even these are fundamentally built upon the same principles of atomic structure. They exist fleetingly, giving us glimpses into the "nuclear drip line" where nuclei simply can't hold together anymore. We've even been able to synthesize a few such as Californium, often used in specialized applications. You can delve deeper into the fascinating world of transuranic elements on Wikipedia.
The Quest for Superheavy Elements: An Earth-bound Endeavor
Scientists on Earth are in a race to create and understand **superheavy elements**—those with atomic numbers far exceeding uranium. The theory suggests an "island of stability" exists around atomic numbers 114, 120, or 126, where certain configurations of protons and neutrons might lead to surprisingly long-lived, albeit still radioactive, isotopes. Imagine an element with over 120 protons, existing not for nanoseconds, but perhaps for minutes, hours, or even days!
The hunt involves smashing lighter nuclei together at incredible speeds, hoping they fuse into something heavier. This is delicate work, like trying to throw two wet tissues at each other and hoping they stick. The challenges are immense: the small cross-section for fusion, the need for precise beam energies, and the sheer difficulty of detecting a handful of atoms that decay almost immediately.
But even if we reach this "island of stability," these are still extensions of our current periodic table, following the same rules of quantum mechanics that govern lighter atoms. They are "superheavy" but not necessarily "impossible" in the sense of fundamentally new types of matter.
Cosmic Forges: Where the Truly Impossible Might Arise
Now, let's shift our gaze to the most extreme environments in the universe. These are places where gravity crushes matter to unimaginable densities, where temperatures soar to billions of degrees, and where exotic particles might interact in ways we can only theorize.
Neutron Stars: Laboratories of Extreme Matter
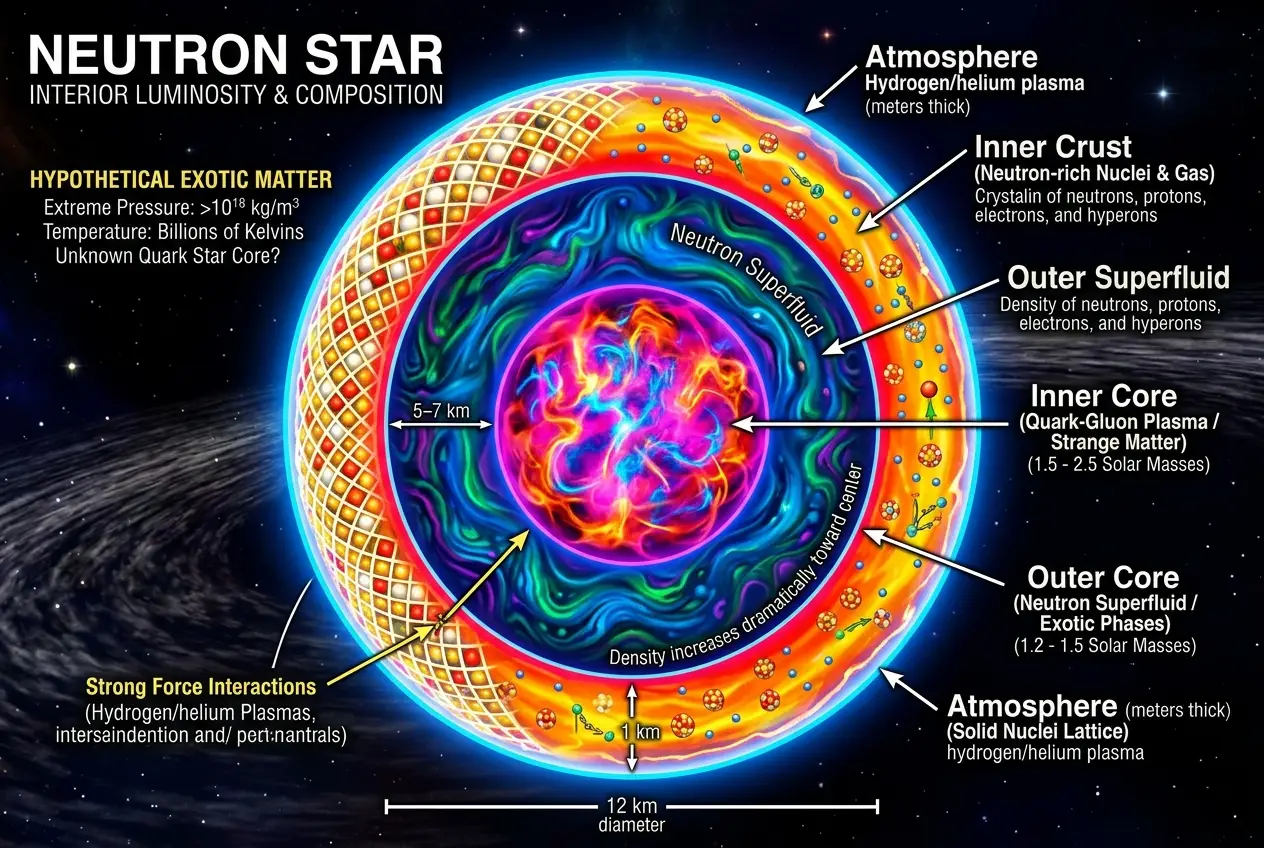
One of the most promising candidates for forging truly exotic matter is the **neutron star**. These stellar remnants are born from the catastrophic collapse of massive stars, packing the mass of our sun into a sphere barely the size of a city. The gravity is so immense that electrons are forced into protons, creating neutrons.
I find it mind-boggling to consider the conditions inside a neutron star. The outer crust might consist of atomic nuclei in a crystalline lattice, but the inner layers are a mystery. We believe they contain a superfluid of neutrons, but deeper still, the pressure could be so great that individual neutrons break down. Could these conditions lead to:
* **Strange Matter:** A hypothetical form of quark matter consisting of roughly equal numbers of up, down, and strange quarks. If stable, strange matter could be denser than neutronium, forming "strangelets" or even entire "strange stars." The idea is fascinating, implying a fundamental rearrangement of matter itself, beyond conventional atoms. You can read more about strange matter theories on Wikipedia.
* **Hyperons:** At such extreme densities, nucleons (protons and neutrons) might convert into other, heavier baryons called hyperons, which contain strange quarks. This would significantly alter the equation of state of matter, making neutron stars even more compact.
* **Elements with Exotic Bonds:** Perhaps elements don't even form in the way we understand them. What if the extreme nuclear forces create transient, super-dense clusters of quarks and gluons that behave like elements, but without distinct protons and neutrons?
Supernovae and Kilonovae: Cosmic Alchemy
Supernovae, the explosive deaths of massive stars, are known to be the primary sites for the **r-process (rapid neutron capture process)**, which creates many of the heavy elements we find on Earth, like gold and platinum. During a supernova, a huge flux of neutrons is produced, which are rapidly absorbed by existing nuclei before they can decay. This quickly builds up heavier and heavier elements.
When two neutron stars collide, an event known as a **kilonova**, the conditions are even more extreme than in a typical supernova. These mergers produce an incredible amount of heavy elements, many times over what a single supernova can achieve. Could this hyper-r-process push the boundaries even further, creating elements so heavy they exist only transiently as part of the ejecta before decaying into known, lighter elements?
I sometimes wonder if the periodic table we have is merely a snapshot of stable (or semi-stable) configurations. What if there's an entire ephemeral zoo of elements that pop into existence during these cosmic cataclysms, only to vanish before we can even begin to detect them? It’s a compelling thought that the most energetic events in the cosmos are essentially fleeting alchemical factories. You can explore the intricacies of nucleosynthesis and the r-process on Wikipedia.
Could We Detect Them? The Challenge of Cosmic Anomalies
Detecting these "impossible" elements or exotic forms of matter presents enormous challenges. If they are extremely short-lived or only stable under specific, intense conditions, how would we ever know they existed?
1. **Gravitational Wave Signatures:** Kilonovae and neutron star mergers are prime sources of gravitational waves. Subtle differences in the gravitational wave signals could potentially reveal insights into the precise nature of the matter involved in the merger, including the creation of exotic elements or strange matter.
2. **Spectroscopic Signatures:** If any such exotic elements could exist in a more "stable" form within the outer layers of a neutron star, or briefly survive in the aftermath of a cosmic explosion, they might emit or absorb light at unique wavelengths. Our powerful telescopes, like the James Webb Space Telescope, could theoretically detect these unique spectral lines. However, predicting what those lines would look like for an element outside our current understanding is a monumental task.
3. **Cosmic Ray Signatures:** High-energy cosmic rays bombard Earth constantly. Some are thought to originate from supernovae. Could a rare, superheavy cosmic ray particle, defying known elemental compositions, be a messenger from a cosmic forge? This is highly speculative, but the idea is thrilling.
4. **Indirect Evidence:** Much like we infer the existence of dark matter through its gravitational effects, we might first find indirect evidence of impossible elements—perhaps through unusual stellar phenomena or unexpected energy releases that can't be explained by known physics.
The search for these cosmic anomalies is inherently intertwined with our broader quest to understand the universe's fundamental laws. What if we are missing a piece of the puzzle because we are only looking for patterns we already recognize? The universe is under no obligation to conform to our textbooks.
Beyond the Periodic Table: What Would "Impossible" Even Mean?
When I use the term "impossible," I'm not suggesting a violation of physics. Instead, I mean elements or forms of matter that exist outside the *current framework* of our periodic table and nuclear models. This could mean:
* **Nuclei with fundamentally different internal structures:** Not just more protons and neutrons, but perhaps different configurations of quarks or even stranger constituents.
* **Elements stable only under extreme conditions:** Much like a diamond is stable under high pressure but turns to graphite otherwise. These elements would be "impossible" to sustain in normal conditions.
* **New forces at play:** What if the immense gravity or unique particle interactions within a neutron star activate a force that governs matter in ways we haven't yet discovered?
The very idea challenges our conception of what an "element" truly is. Is it defined solely by the number of protons and neutrons, or could it be a more fluid concept in the cosmic melting pots of space? For further reading on the frontiers of cosmic element formation, you might find this article on the formation of heavy elements in the universe insightful.
| Element Category |
Characteristics |
Typical Cosmic Origin |
| Light Elements (H, He, Li) |
Formed in the Big Bang. Essential building blocks. |
Big Bang Nucleosynthesis |
| Common Heavy Elements (C-Fe) |
Stable, fundamental for planets and life. |
Stellar Nucleosynthesis (fusion in stars) |
| Superheavy Elements (Z > 92) |
Highly unstable, radioactive. Some theorized "island of stability." |
Supernovae (r-process), Kilonovae, Lab Synthesis |
| Hypothetical "Impossible" Elements |
Stable only under extreme pressure/temperature, or new forms of quark matter. |
Neutron Star Interiors, Early Universe Conditions |
The Endless Frontier of Cosmic Discovery
The universe is a place of profound complexity and wonder, constantly challenging our preconceptions. The very idea that "impossible" elements might be hiding in space is a testament to this. It reminds us that our scientific models, while powerful, are always evolving, always pushing towards a more complete picture of reality.
As we continue to explore the cosmos with ever more sophisticated instruments, from advanced gravitational wave detectors to next-generation telescopes, I believe we stand on the cusp of discoveries that could completely rewrite our understanding of matter. The next time you look up at the stars, consider not just the familiar elements that make up our world, but also the potential for something entirely new, forged in the universe's most violent and mysterious corners. It's a humbling thought that the universe might still hold secrets about the very fabric of existence, waiting for us to uncover them. For more on the mysteries of space, you might enjoy reading about whether /blogs/is-planet-nine-a-primordial-black-hole-8612 or if /blogs/do-black-holes-store-universes-lost-data-5797.

 Join Us
Join Us





































Leave a Reply
Comments (0)