The first time I saw a high-resolution image of Jupiter's Great Red Spot, I was mesmerized. It wasn't just a storm; it was a cosmic eye, swirling with unfathomable power, large enough to swallow Earth whole, and ancient beyond human comprehension. It’s been raging for centuries, a persistent anomaly on the largest planet in our solar system. For decades, scientists have peered into its fiery depths, trying to understand its longevity and the complex dynamics of Jupiter's atmosphere. But what if this iconic tempest isn’t just a maelstrom of gas and vapor? What if, hidden beneath its crimson swirls and raging winds, lies an unimaginable treasure: **oceans of liquid diamond?**
It sounds like something straight out of a science fiction novel, a glittering, high-pressure world of precious gems. Yet, the extreme conditions on gas giants like Jupiter push the boundaries of known physics and chemistry, creating environments where the ordinary becomes truly extraordinary. Could Jupiter, our solar system's colossal guardian, truly harbor such a dazzling secret? Let's dive into the fascinating science and tantalizing possibilities that suggest this cosmic fantasy might just be a reality.
## The Great Red Spot: A Vortex Beyond Earthly Comprehension
For centuries, astronomers have observed the Great Red Spot (GRS) on Jupiter, a persistent high-pressure region that has been recorded for at least 350 years, and possibly much longer. It's an anticyclonic storm, meaning it rotates counter-clockwise in Jupiter's southern hemisphere, and it’s truly enormous. At its largest, it was wide enough to fit three Earths side-by-side, though it has been shrinking over time. The winds within the GRS howl at speeds exceeding 400 miles per hour, generating turbulence that we can barely imagine on our home planet. For more fascinating details on this cosmic behemoth, you can explore its history on [Wikipedia's Great Red Spot page](https://en.wikipedia.org/wiki/Great_Red_Spot).
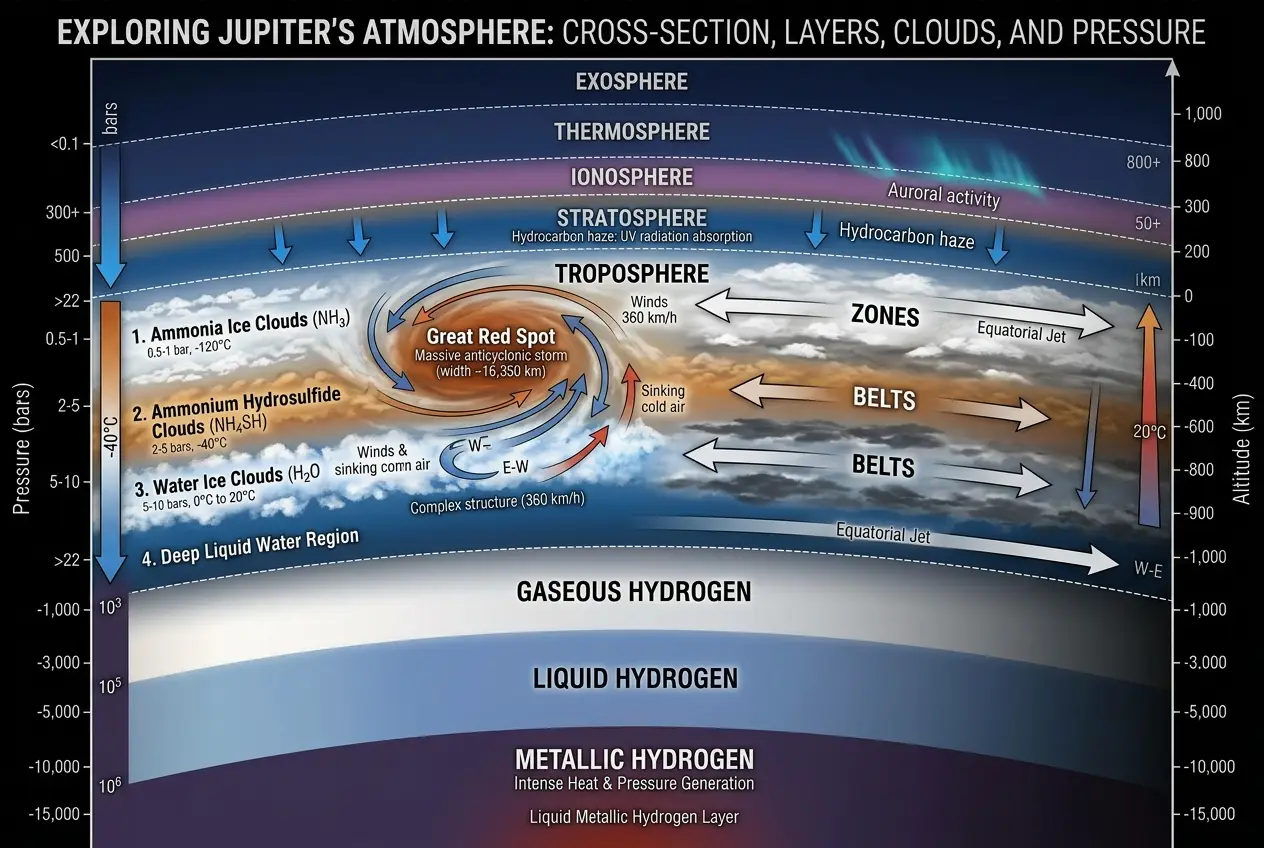
Jupiter's atmosphere itself is a chaotic ballet of hydrogen, helium, and trace amounts of methane, ammonia, and water vapor. As you descend into its interior, the pressure and temperature increase dramatically. Hydrogen, which is a gas at the top, transforms into a liquid metal under immense pressure, capable of conducting electricity. It's a world fundamentally different from our rocky Earth, where exotic states of matter are not just theoretical but are expected to exist.
## Beneath the Clouds: The Chemistry of Extreme Pressure
To understand how diamonds might form, we first need to consider the composition of Jupiter. While hydrogen and helium dominate, there are also significant amounts of carbon-containing compounds, primarily methane (CH₄). On Earth, carbon forms diamonds under extreme heat and pressure, typically deep within the planet's mantle. But Jupiter isn't Earth. It's a gas giant, a massive orb with a crushing gravitational pull that generates pressures far exceeding anything found in Earth's crust.
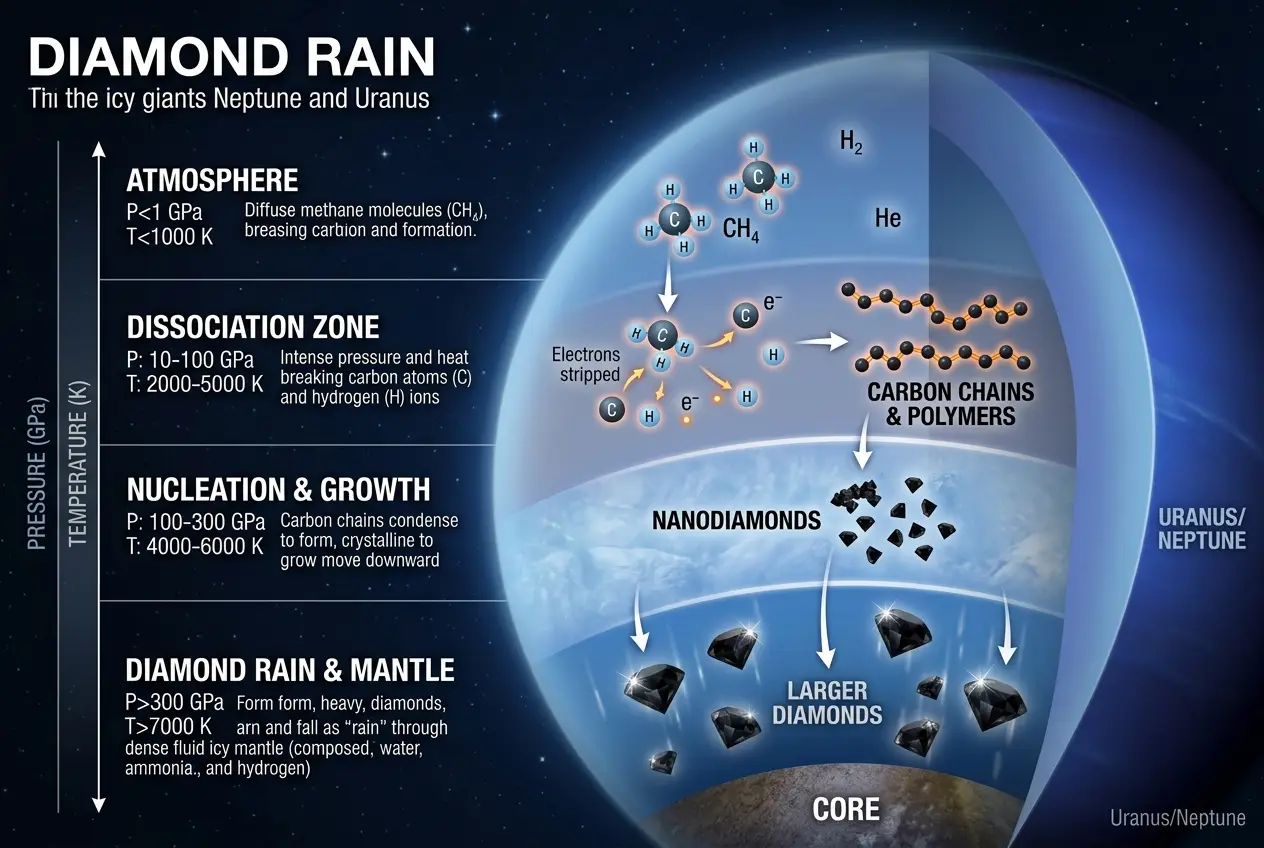
Scientists have long hypothesized that diamond rain falls on other ice giants like Neptune and Uranus. These planets have a higher proportion of methane in their atmospheres compared to Jupiter. Under the immense pressures and temperatures in their interiors, methane molecules can be ripped apart. The carbon atoms then precipitate, forming solid diamonds that slowly sink toward the core, often described as "diamond rain." I've been fascinated by the idea of extreme material formation, and this concept isn't entirely new. We've explored how such incredible forces can forge new materials for future technology in a previous blog post about how [pressure could forge unseen materials](/blogs/could-pressure-forge-unseen-materials-for-future-tech-7524).
The process generally involves:
1. **Methane breakdown:** Extreme heat from lightning or atmospheric dynamics breaks down methane (CH₄) into elemental carbon and hydrogen.
2. **Carbon Condensation:** Under enormous pressure, these carbon atoms condense into solid diamond chunks.
3. **Gravitational Descent:** These diamonds, denser than the surrounding hydrogen-helium fluid, fall through the atmosphere.
This exact phenomenon has even been recreated in laboratories, using powerful lasers to subject polystyrene (a carbon-rich plastic) to the pressures found within Neptune, producing nanodiamonds. This fascinating experiment provides tangible evidence that diamond formation under such conditions is not just theoretical.
## Could Jupiter Join the Diamond Club?
Jupiter, being mostly hydrogen and helium, has less methane than Uranus or Neptune. However, it still contains a non-negligible amount, and its sheer size means that even small percentages translate into vast quantities of material. The key question for Jupiter is whether the conditions necessary for diamond formation—sufficient carbon, high enough pressure, and the right temperature range—exist.
The Great Red Spot itself might be a critical factor. The tremendous energy of this storm generates powerful lightning, which can be thousands of times more energetic than Earth’s lightning. This intense electrical activity can break down methane molecules in the upper atmosphere, creating soot or graphite particles. As these carbon particles descend deeper into Jupiter's atmosphere, the pressures and temperatures increase rapidly. Scientists speculate that at depths where pressures are millions of times greater than Earth's sea level, these carbon atoms could be compressed into solid diamonds.
But could these diamonds form *oceans*? This is where the idea truly sparkles. On Earth, diamonds are solid. However, under the mind-boggling pressures and temperatures found deep within Jupiter, the properties of matter change dramatically. Just as ice can melt into water, and water can become supercritical fluid, solid diamonds might melt into a liquid form of carbon under extreme conditions.
"The phase diagram of carbon is exceptionally complex," says Dr. Kevin Baines from NASA's Jet Propulsion Laboratory, whose research has explored the possibility of diamond rain on gas giants. "Under the right temperatures and pressures, you can move from graphite to diamond, and then even to liquid carbon."
This concept of liquid carbon, or perhaps a slushy mixture of solid diamonds suspended in liquid carbon, creates the dazzling vision of diamond oceans. These "oceans" wouldn't be like Earth's water bodies; they would be incredibly dense, super-hot, and under pressures that would instantly crush anything we know. Imagine a liquid so dense and hot that it glows, constantly churning with solid diamond fragments. It's a truly alien seascape. We've even discussed other planetary diamond phenomena in our blog post about [diamond rain and cosmic superconductors](/blogs/diamond-rain-can-planets-forge-cosmic-superconductors-6917).
## Juno's Glimpses and Future Explorations
The Juno spacecraft, currently orbiting Jupiter, has provided unprecedented insights into the planet’s gravity and magnetic fields, allowing scientists to infer details about its deep interior structure. While Juno can't directly "see" a diamond ocean, its data helps refine models of Jupiter's composition and the state of matter within its layers. The mission has already revealed surprising details about Jupiter's deep atmosphere and its powerful magnetic field, which is generated by the movement of its metallic hydrogen interior. For more information on Jupiter's complex structure, the [Wikipedia page on Jupiter's interior](https://en.wikipedia.org/wiki/Jupiter#Interior) offers excellent details.
Proving the existence of diamond oceans or even diamond rain on Jupiter is an immense challenge. It would require probes capable of withstanding unimaginable pressures and temperatures, descending far deeper than any spacecraft has gone before. Such missions are still decades away, requiring revolutionary materials and propulsion systems. Until then, our understanding relies on theoretical models, laboratory experiments that simulate extreme conditions, and the indirect data gathered by missions like Juno.
The scientific journey to uncover these secrets continues to fuel our curiosity about the universe. From trying to understand if [some planets sing in radio waves](/blogs/why-do-some-planets-sing-in-radio-waves-4437) to the ongoing hunt for a hypothetical Planet Nine, the cosmos constantly challenges our perceptions and invites us to dream bigger.
## The Broader Implications
The possibility of diamond oceans on Jupiter is more than just a captivating idea; it pushes the boundaries of our understanding of planetary formation and the exotic chemistry possible in the universe. If such conditions exist, it suggests that valuable resources could be locked away on worlds across the galaxy, altering our long-term perspectives on interstellar resource gathering. More fundamentally, it highlights the incredible diversity and strangeness of the cosmos, reminding us that nature's imagination often far exceeds our own.

While the direct confirmation of Jupiter's diamond oceans remains a challenge for future generations of space explorers, the scientific principles supporting its possibility are firmly rooted in high-pressure physics and planetary chemistry. It's a testament to the fact that even in our own cosmic backyard, there are still astounding secrets waiting to be unearthed, reminding us that the universe is far wilder and more wonderful than we can often conceive. The next time you gaze up at the night sky and spot Jupiter, remember the possibility of glittering, liquid diamonds swirling deep within its crimson eye.
Frequently Asked Questions
Currently, there is no direct observational evidence of diamond oceans on Jupiter. The idea is based on theoretical models of extreme planetary interiors, laboratory experiments simulating high-pressure conditions, and indirect data from missions like Juno, which help us understand Jupiter's internal structure and composition.
Under the incredibly immense pressures and temperatures deep within Jupiter, carbon atoms, which form solid diamonds on Earth, could transition into a liquid state. This is similar to how water can exist as a gas, liquid, or solid, or even a supercritical fluid, depending on pressure and temperature. High-pressure physics suggests that solid diamonds would melt into liquid carbon under such extreme conditions.
The Great Red Spot is a powerful storm that generates intense lightning, far more energetic than Earth's. This lightning could play a crucial role by breaking down methane (a carbon-containing compound) in Jupiter's upper atmosphere into elemental carbon. As these carbon particles descend deeper into the planet, they could then be subjected to the pressures and temperatures necessary to form diamonds, and potentially liquid carbon.
While the concept of *liquid* diamond oceans is a more extreme hypothesis for Jupiter, the idea of 'diamond rain' – where solid diamonds precipitate – is more widely accepted for ice giants like Neptune and Uranus due to their higher methane content. Jupiter's unique combination of immense size, internal heat, and atmospheric dynamics could potentially lead to even more exotic carbon phases like liquid diamonds.
Scientists use specialized equipment like diamond anvil cells or powerful lasers to create conditions of extreme pressure and temperature in laboratories. By compressing and heating carbon-rich materials, they can simulate the environments found within gas giants and observe the formation of nanodiamonds, supporting the theoretical models for diamond precipitation on these distant worlds.
Verified Expert
Alex Rivers
A professional researcher since age twelve, I delve into mysteries and ignite curiosity by presenting an array of compelling possibilities. I will heighten your curiosity, but by the end, you will possess profound knowledge.
Leave a Reply
Comments (0)
No approved comments yet. Be the first to share your thoughts!
Leave a Reply
Comments (0)