I once saw a parlor trick that completely blew my mind. Someone held a small, silvery spoon, seemingly made of metal, and placed it into a cup of warm tea. Within seconds, the spoon began to droop, then sag, and finally, melted away into a shimmering puddle at the bottom of the cup. My first thought was, “Is this magic? Or some elaborate hoax?” But the truth, as I soon learned, was far more fascinating than any illusion. It was a demonstration of the peculiar properties of certain metals – metals that defy our everyday understanding by melting at temperatures we consider ‘room temperature’ or just slightly above.
We're taught that metals are strong, solid, and require immense heat to turn into a liquid. Think of steel forged in a furnace or gold melted down for jewelry. Yet, the world of materials science harbors anomalies that challenge these preconceptions, hinting at a universe of possibilities for future technology. These low-melting-point metals aren't just curiosities; they represent a frontier in electronics, soft robotics, and even advanced thermal management.
### The Science Behind the Melt: Intermolecular Forces and Atomic Structure
At its core, the melting point of a substance is determined by the strength of the forces holding its atoms or molecules together. For metals, these are primarily **metallic bonds** – a "sea" of delocalized electrons shared among a lattice of positive metal ions. The stronger these bonds, the more energy (and thus higher temperature) is required to break the rigid crystalline structure and allow the atoms to move freely, transitioning from solid to liquid.
So, why do some metals require so little energy to achieve this state? It boils down to a combination of factors related to their atomic structure and electronic configurations:
* **Weak Metallic Bonds:** Some metals have relatively weaker metallic bonds. This can be due to a larger atomic size, which means the valence electrons are further from the nucleus and less strongly attracted, or a lower number of valence electrons contributing to the "electron sea."
* **Unique Crystal Lattices:** The specific arrangement of atoms in a metal's crystal lattice also plays a crucial role. Some structures are inherently less stable and require less energy to dislodge atoms from their fixed positions. For instance, the atomic structure of gallium makes it quite "floppy" compared to other metals.
* **Electron Shell Effects:** Certain elements, particularly those with filled or nearly filled d-orbitals, exhibit weaker metallic bonding due to relativistic effects that impact how electrons interact. This is part of the reason elements like mercury are liquid at room temperature.
### Gallium: The Hand-Melting Metal
The most famous example, and likely what I witnessed, is **gallium (Ga)**. With a melting point of just 29.76 °C (85.57 °F), it melts readily in the warmth of a human hand. Gallium is a soft, silvery-blue metal that solidifies into a brittle solid, but a slight increase in temperature transforms it into a mirror-like liquid. Its unique properties make it a darling of scientific demonstrations and a promising material for various high-tech applications.
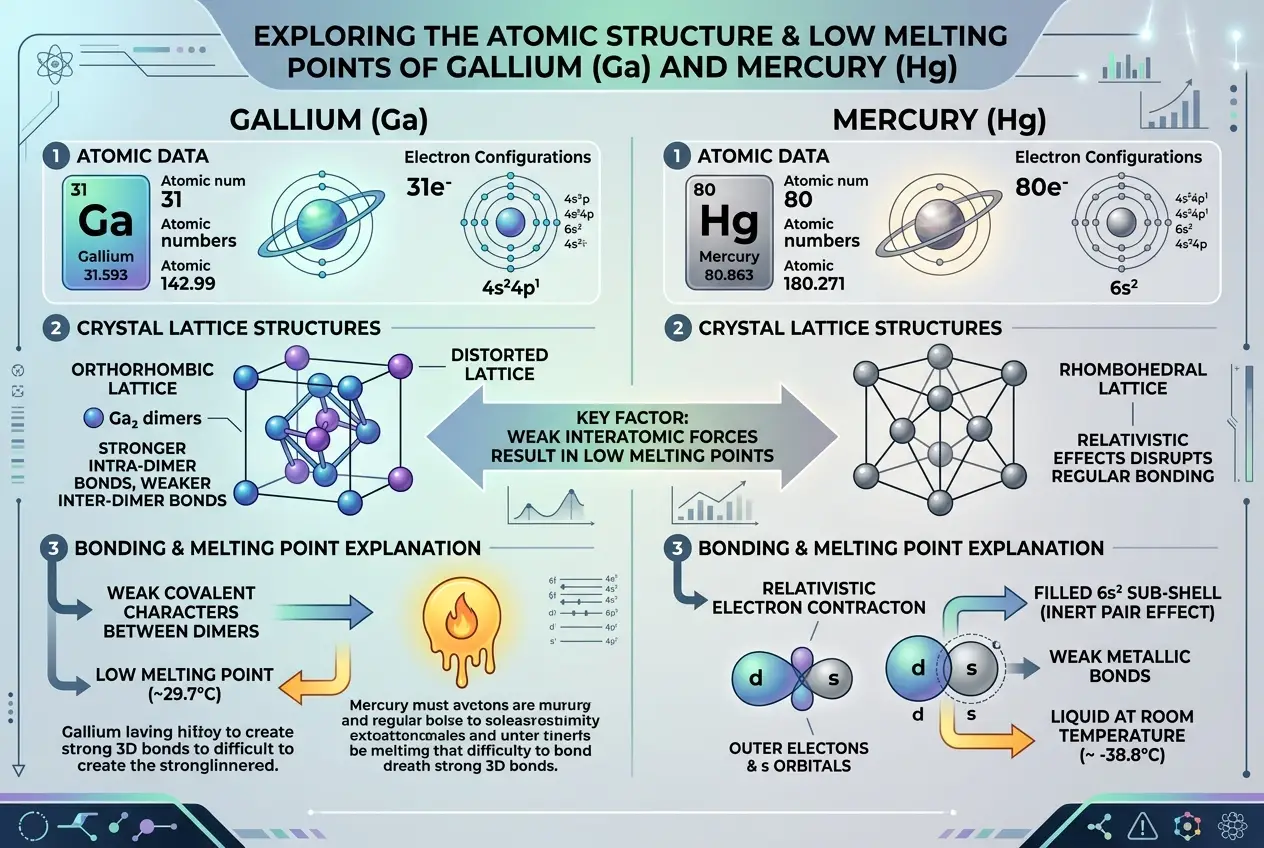
Gallium's low melting point is primarily attributed to its unusual crystal structure in the solid phase. Unlike many metals that form simple cubic or hexagonal close-packed structures, solid gallium forms a unique orthorhombic crystal with diatomic Ga₂ units. The bonds *between* these Ga₂ units are relatively weak, requiring less energy to break them and transition to a liquid state where the atoms can move more freely. This contrasts sharply with stronger metallic bonds in common metals like iron or copper, which remain solid at much higher temperatures. For a deeper dive into gallium's properties, explore its Wikipedia page [here](https://en.wikipedia.org/wiki/Gallium).
### Beyond Gallium: Other Low-Melting Wonders
While gallium often steals the spotlight, it's not the only metal with such intriguing thermal properties.
* **Mercury (Hg):** The classic example, mercury, is liquid at room temperature with a melting point of -38.83 °C (-37.89 °F). Its uniqueness stems from relativistic effects that cause its 6s electrons to be bound more tightly to the nucleus, making them less available for metallic bonding. This weakens the overall metallic bond strength, resulting in its liquid state.
* **Cesium (Cs):** With a melting point of 28.4 °C (83.1 °F), cesium is another metal that can melt in warm ambient conditions. It's an alkali metal, known for its large atomic size and single valence electron, leading to very weak metallic bonding. However, its extreme reactivity with air and water makes it less practical for casual demonstrations than gallium.
* **Rubidium (Rb):** Close behind cesium, rubidium melts at 39.3 °C (102.7 °F). Like cesium, it's highly reactive and shares similar reasons for its low melting point: large atomic size and weak metallic bonds.
* **Indium (In):** Melting at 156.6 °C (313.9 °F), indium isn't quite a "room temperature" melter, but its low melting point for a metal makes it valuable in various applications, particularly in solders and low-temperature alloys.
### The Dawn of Liquid Tech: Applications and Innovations
The peculiar properties of low-melting-point metals are far from mere scientific curiosities. They are actively being explored and integrated into groundbreaking technologies that promise to reshape our future:
#### 1. Flexible and Stretchable Electronics
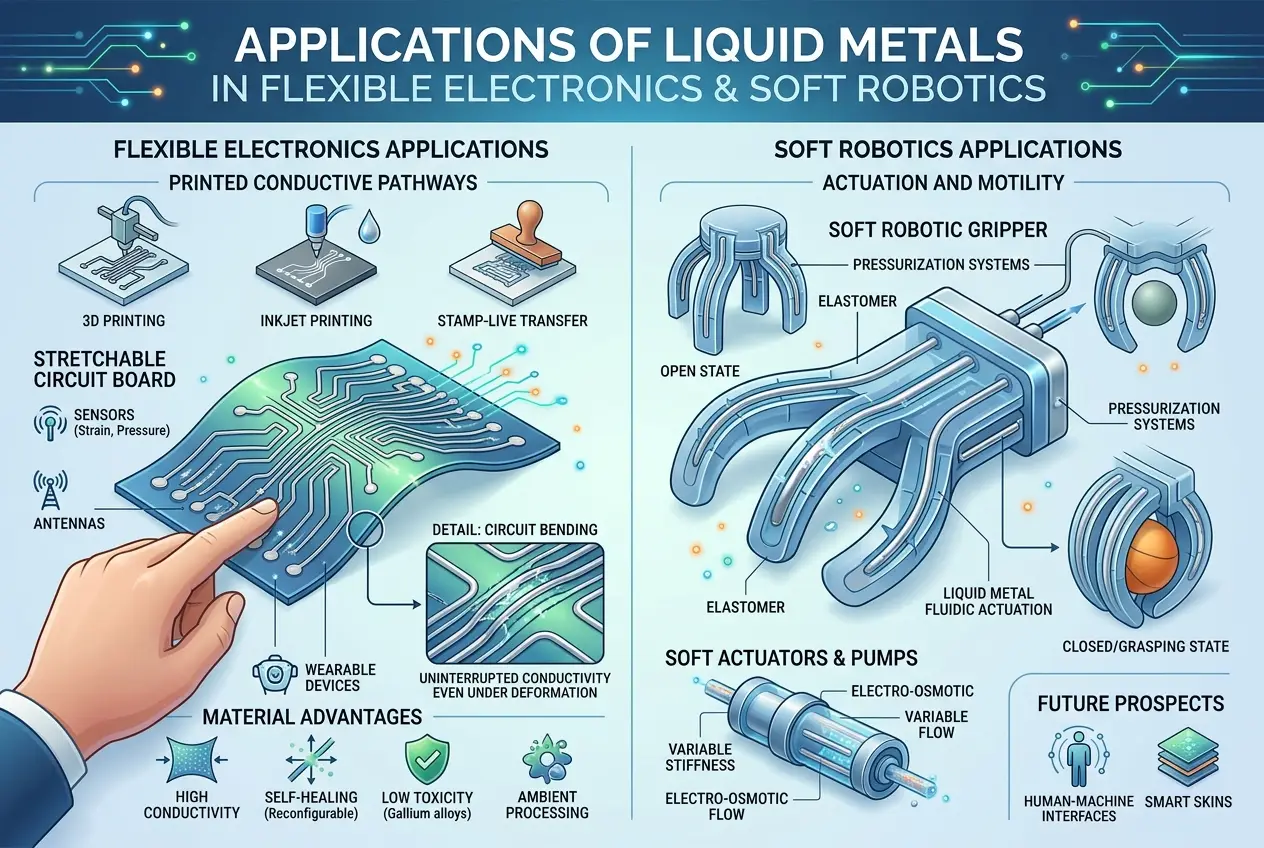
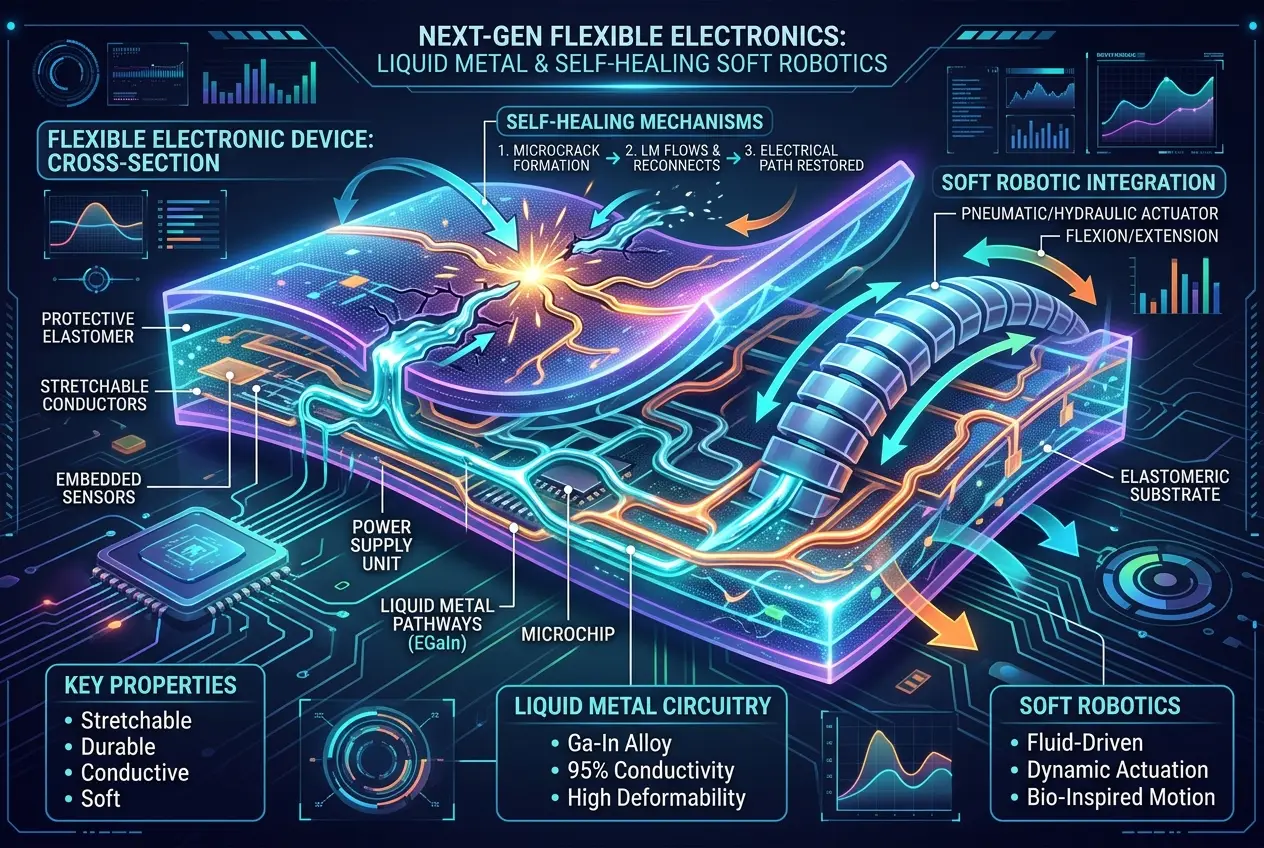
Imagine electronic circuits that can bend, stretch, and even heal themselves. Metals like gallium and its alloys (e.g., galinstan, an alloy of gallium, indium, and tin) are excellent candidates for this. Their liquid state at room temperature allows them to be encapsulated within flexible polymers, forming conductive pathways that can withstand significant deformation without breaking. This is crucial for wearables, implantable medical devices, and soft robotics. Researchers are actively developing techniques to print liquid metal circuits, paving the way for truly flexible displays and self-repairing wires. You can learn more about flexible electronics and their potential impact on our daily lives in our blog post on [Can Living Cells Build Our Next Supercomputers?](/blogs/can-living-cells-build-our-next-supercomputers-6472).
#### 2. Soft Robotics
Traditional robots are often rigid and metallic, designed for precision but lacking in dexterity and adaptability. Soft robotics aims to create robots that can safely interact with humans and navigate complex environments. Liquid metals are game-changers here. They can be used to create deformable actuators, sensors, and power sources that mimic biological systems, allowing robots to change shape, grip delicate objects, and even move like natural organisms. The ability to flow and solidify on demand opens up possibilities for self-healing robotic components.
#### 3. Advanced Thermal Management
Electronic devices generate heat, and managing this heat is critical for performance and longevity. Liquid metals, particularly their alloys, boast excellent thermal conductivity – often far superior to traditional thermal pastes or air cooling. This makes them ideal for cooling high-performance processors in computers, servers, and even next-generation fusion reactors. Their ability to flow and adapt to complex surfaces ensures efficient heat transfer, pushing the boundaries of what's possible in compact, powerful electronics.
#### 4. Self-Healing Materials
Cracks and damage in materials are a common problem. Liquid metals offer a fascinating solution through self-healing mechanisms. Imagine a material designed with microchannels filled with liquid metal. If a crack forms, the liquid metal flows into the void, restoring conductivity or structural integrity. This could extend the lifespan of components in everything from spacecraft to everyday consumer electronics, reducing waste and maintenance costs. The concept of self-healing materials echoes some of the incredible durability of ancient structures discussed in our blog, [Roman Concrete: Did Ancients Engineer Self-Healing Tech?](/blogs/roman-concrete-did-ancients-engineer-self-healing-tech-8467).
#### 5. Catalysis and Chemical Reactions
The unique surface properties of liquid metals can also make them effective catalysts for various chemical reactions. Their high surface tension and ability to be easily manipulated provide new platforms for chemical synthesis, offering greener and more efficient pathways for industrial processes. Research into liquid metal catalysts is a growing field, promising innovations in energy production and environmental clean-up. You can find more information about the use of liquid metals in catalysis on this [Wikipedia page](https://en.wikipedia.org/wiki/Liquid_metal#Catalysis_and_Chemical_Reactions).
### The Challenges and the Future
While the potential of low-melting-point metals is immense, challenges remain. Reactivity with other materials, toxicity concerns (especially with mercury), and the complexity of integrating them into large-scale manufacturing processes are hurdles that researchers are actively addressing. For instance, developing stable encapsulations for liquid metal circuits and exploring less toxic alternatives are key areas of focus.
Despite these challenges, the future of "liquid tech" looks incredibly bright. As our understanding of materials science deepens and our engineering capabilities advance, we are likely to see these curious metallic liquids move from laboratory demonstrations to indispensable components in the next generation of smart devices, adaptable robots, and efficient energy systems. The metals that melt in your hand might just be the building blocks of tomorrow's most revolutionary innovations.
The world of materials is full of surprises, constantly revealing new possibilities that defy our conventional wisdom. The simple act of a metal dissolving in a cup of tea is a powerful reminder that sometimes, the most peculiar phenomena hold the key to unlocking extraordinary technological advancements.
Frequently Asked Questions
While some, like mercury, are toxic and require careful handling, others like gallium are relatively non-toxic. Research focuses on encapsulating these metals and developing safe alloys for commercial applications, especially in consumer electronics and medical devices.
Engineers employ various strategies, including encapsulation within inert polymers, creating alloys that reduce reactivity, and designing systems where liquid metals are isolated from corrosive environments. Surface coatings and specialized packaging also play a crucial role.
While some metals naturally have low melting points due to their atomic structure, scientists can create alloys (mixtures of metals) that have significantly lower melting points than their individual components. Eutectic alloys, for example, melt at a lower temperature than any of their constituents.
Mercury holds the record among pure metals, melting at -38.83 °C. However, certain metal alloys, particularly some eutectic alloys, can have even lower melting points, with some experimental mixtures reported to remain liquid at temperatures below -50 °C.
Absolutely! Their excellent thermal conductivity makes them ideal for cooling spacecraft components or managing heat in extreme space environments. Their ability to form flexible structures could also be beneficial for deployable antennas or adaptive robotic tools on other planets.
Verified Expert
Alex Rivers
A professional researcher since age twelve, I delve into mysteries and ignite curiosity by presenting an array of compelling possibilities. I will heighten your curiosity, but by the end, you will possess profound knowledge.
Leave a Reply
Comments (0)
No approved comments yet. Be the first to share your thoughts!
Leave a Reply
Comments (0)