I recently came across an idea that stopped me in my tracks. Imagine a battery, not powered by unstable chemicals or limited by charge cycles, but by something as eternal and enduring as a diamond. What if these diamonds weren't just jewelry, but the ultimate power source, capable of lasting thousands of years, all while cleaning up one of humanity’s most pressing environmental challenges? This isn't science fiction anymore; it’s the intriguing, complex world of **diamond batteries**, a concept that promises to redefine our relationship with energy and nuclear waste.
The global energy demand is relentless, pushing us to seek cleaner, more sustainable alternatives. Solar, wind, and geothermal have their place, but they often face issues of intermittency and storage. Meanwhile, nuclear power, while powerful, leaves behind a legacy of highly radioactive waste that demands secure, long-term disposal. But what if we could turn that very waste into a solution, rather than a problem? This is where the groundbreaking potential of diamond batteries enters the picture.
## The Genesis of a Brilliant Idea: Nuclear Waste as a Power Source
The concept of a diamond battery isn't about gem-encrusted power packs for your smartphone (though that would be quite a flex!). It stems from a fascinating field called **betavoltaics**. Unlike traditional solar cells that convert light, betavoltaic devices convert beta decay electrons (a type of radiation) into electrical current. Think of it as a miniature nuclear generator, but one that is completely safe and contained.
The brilliant twist comes from using **radioactive isotopes** like Carbon-14 (C-14), Nickel-63 (Ni-63), or Tritium (H-3), which are byproducts of nuclear fission in graphite moderators from nuclear reactors. Specifically, C-14 is particularly abundant in the graphite blocks used in older Magnox reactors. These materials have half-lives ranging from decades to thousands of years, meaning they emit low levels of radiation for an incredibly long time. The challenge has always been how to safely harness this energy without the radiation becoming a hazard.
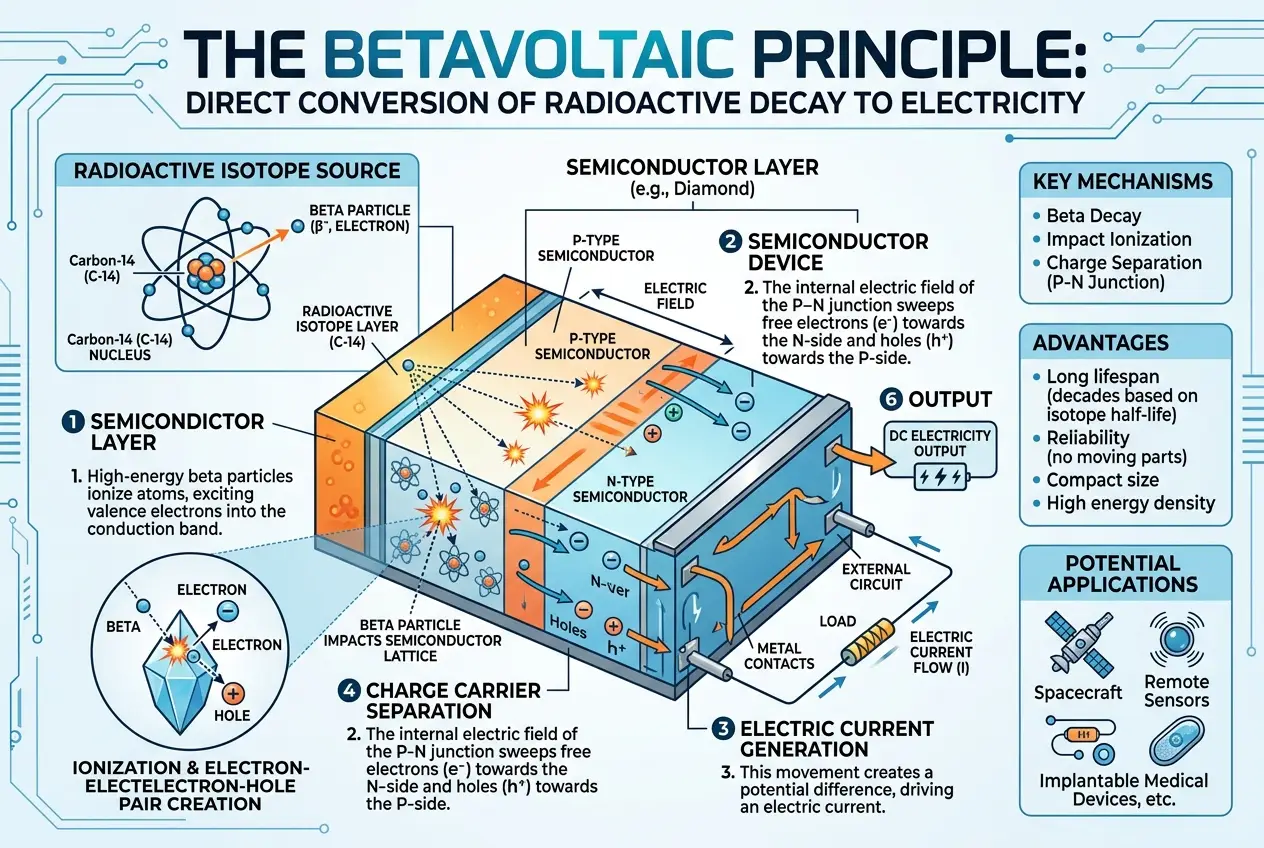
Researchers, notably at the University of Bristol in the UK, have pioneered the idea of embedding these radioactive isotopes *inside* artificial diamonds. Diamonds are exceptional materials: incredibly hard, excellent thermal conductors, and, crucially, very effective semiconductors. When a beta particle (an electron) emitted by the decaying isotope collides with the carbon atoms in the diamond lattice, it creates electron-hole pairs, generating a small but continuous electric current. The diamond effectively acts as both the radiation source containment and the semiconductor converter.
One of the primary benefits is the *safety* aspect. The diamond itself encapsulates the radioactive material, preventing leakage of harmful radiation. Beta particles are low-energy and easily stopped; even a thin layer of diamond is enough to contain them. This makes the external surface of the battery completely safe to touch, with no detectable radiation.
## Unpacking the Science: How Betavoltaics Work
To understand how these dazzling power cells function, let's delve into the core principles.
A betavoltaic device consists of two main components:
1. **The radioactive source:** This is the material containing the isotope that undergoes beta decay, releasing high-energy electrons. In the case of diamond batteries, this would be a thin layer of C-14 or Ni-63.
2. **The semiconductor converter:** This material absorbs the energy from the beta particles and converts it into electricity. Diamond is an ideal choice due to its wide bandgap, radiation hardness, and high electron mobility.
When a beta particle strikes the diamond lattice, it knocks electrons free from their atomic bonds, creating an electron-hole pair. An internal electric field within the diamond (formed by doping, much like in a solar cell) then sweeps these electrons and holes in opposite directions, generating an electrical current.
**Key advantages of this approach include:**
* **Extreme Longevity:** Imagine a power source that could last for over 5,000 years, requiring no recharging or maintenance. This is the promise of using isotopes with long half-lives. Carbon-14, for instance, has a half-life of 5,730 years.
* **High Energy Density (over time):** While the power output at any single moment might be low, the cumulative energy generated over thousands of years is immense.
* **Waste Recycling:** This technology offers a potential solution for managing radioactive waste, transforming a dangerous byproduct into a valuable resource.
* **Temperature Insensitivity:** Unlike chemical batteries, betavoltaic devices operate efficiently across a wide range of temperatures.
For a deeper dive into the science of betavoltaics, you can explore its principles on [Wikipedia's betavoltaics page](https://en.wikipedia.org/wiki/Betavoltaics).
## Applications Beyond Our Wildest Dreams
So, if these batteries produce low power, where would they be useful? Their unparalleled longevity and stability make them perfect for niche applications where replacing or recharging batteries is impossible or extremely difficult.
### Space Exploration
Think about deep-space probes like Voyager or Cassini. They rely on radioisotope thermoelectric generators (RTGs), which are powerful but use more dangerous isotopes like Plutonium-238. Diamond batteries, with their safer, contained radiation, could power sensors, communications systems, and even entire small spacecraft for centuries, reaching farther into the cosmos than ever before. Imagine a sensor package on a distant exoplanet, quietly gathering data for millennia. This could open doors to exploring even the most remote corners of our solar system and beyond, offering a new perspective on topics like those discussed in [Do Rogue Black Holes Threaten Our Galaxy?](blogs/do-rogue-black-holes-threaten-our-galaxy-6767)
### Medical Implants
Pacemakers, cochlear implants, and other critical medical devices often require battery replacement surgeries every few years. A diamond battery could potentially power these devices for a patient's entire lifetime, reducing surgical risks and improving quality of life. The tiny size and radiation-shielded nature make them incredibly appealing for such sensitive applications.
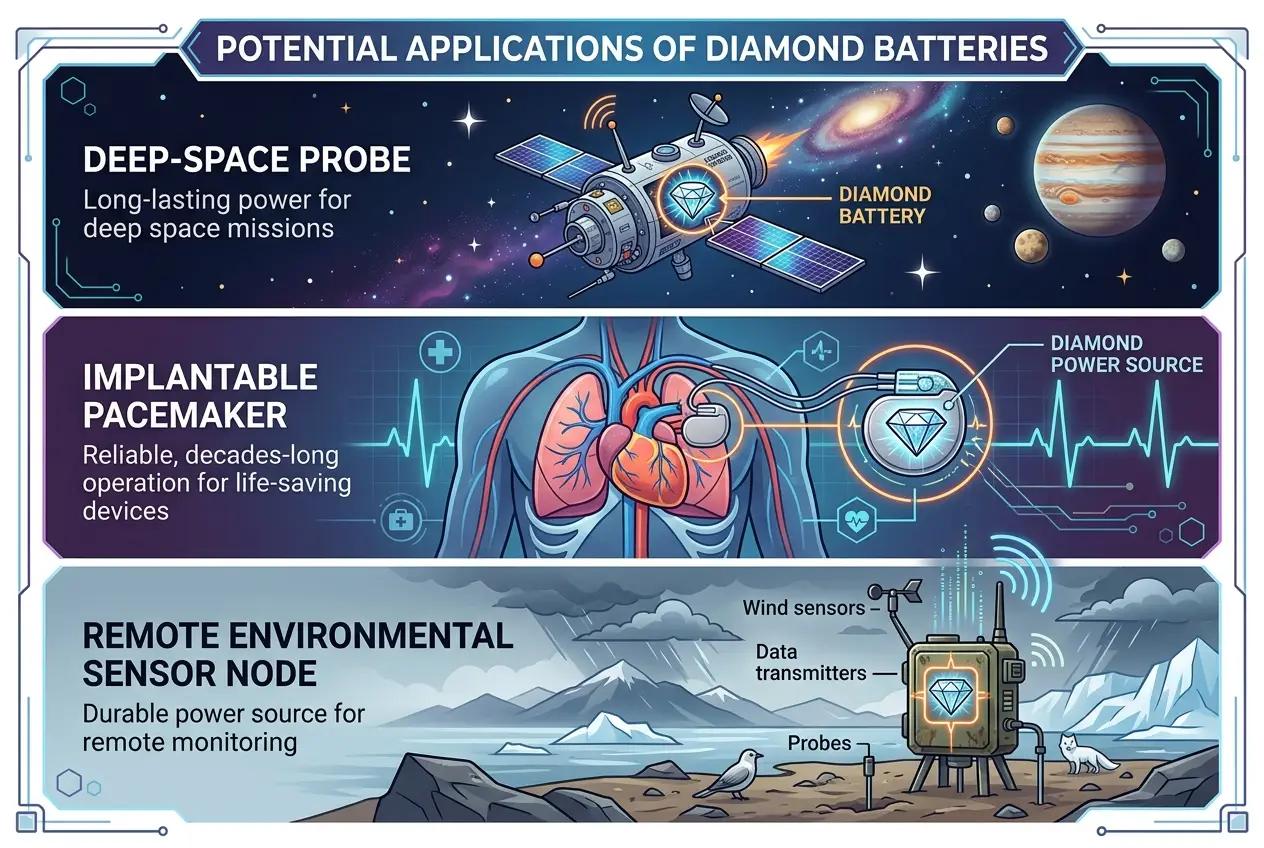
### Remote Sensors and Infrastructure
Monitoring environmental changes in remote areas, powering underwater sensors, or maintaining critical infrastructure in harsh, inaccessible locations—these are all areas where diamond batteries could shine. Imagine bridge sensors that never need battery changes, providing continuous data for hundreds of years. Or geological sensors tracking seismic activity in desolate regions, sending data back for generations. This silent, persistent power source could transform how we gather long-term data, much like the discussions around [Zero-Point Energy: Is Space a Hidden Powerhouse?](blogs/zero-point-energy-is-space-a-hidden-powerhouse-3639) when considering unconventional energy sources.
### Consumer Electronics? Not Quite Yet...
While the allure of a smartphone powered for life is strong, current diamond battery technology delivers very low power output—microwatts to milliwatts. This is far from what modern consumer electronics demand. They are designed for *longevity and reliability*, not high instantaneous power. However, as the technology evolves, efficiency improves, and smaller devices require less power, who knows what the distant future might hold.
## Challenges on the Path to a Diamond-Powered Future
Despite their dazzling potential, diamond batteries face significant hurdles:
* **Low Power Output:** As mentioned, the instantaneous power generated is very small. For many applications, this means diamond batteries would need to be paired with capacitors to store and release bursts of energy, or used in tandem with other energy harvesting methods.
* **Cost of Production:** Creating artificial diamonds and safely embedding radioactive isotopes is currently an expensive and complex process. Scaling up production efficiently is a major challenge.
* **Regulatory Hurdles:** Dealing with radioactive materials, even in a safely encapsulated form, involves strict regulations and public perception issues. Gaining widespread acceptance and approval will require rigorous testing and transparent communication. More information on nuclear waste management can be found on [Wikipedia's article on radioactive waste](https://en.wikipedia.org/wiki/Radioactive_waste).
* **Efficiency:** Converting beta decay directly into electricity is not 100% efficient. Ongoing research aims to improve the conversion efficiency of these betavoltaic devices.
## The Future is Brilliance
The journey from nuclear waste to diamond batteries is a testament to human ingenuity. It’s a remarkable fusion of materials science, nuclear physics, and engineering that offers a tantalizing glimpse into a sustainable future. I believe this technology, even if initially limited to niche applications, represents a profound shift in how we view energy. It transforms a legacy pollutant into a perpetual power source, adding another fascinating dimension to our quest for innovative energy solutions, akin to exploring whether [Could Antimatter Power Our Future?](blogs/could-antimatter-power-our-future-8364).
While we may not be charging our phones with diamonds tomorrow, the underlying science and the audacious vision behind diamond batteries are undeniably captivating. They challenge us to rethink waste, embrace radical longevity, and perhaps, unlock a truly brilliant future for energy. The promise of millennia-long power, derived from the very byproducts we seek to eliminate, is a narrative I'm eager to watch unfold.
Frequently Asked Questions
Diamond batteries are designed to be extremely safe. The radioactive isotopes are encapsulated within the synthetic diamond structure, which acts as a robust shield, preventing any external radiation leakage and making them safe to handle.
Currently, diamond batteries produce very low power output, typically in the microwatt to milliwatt range. They are ideal for applications requiring continuous, low-power energy over extremely long durations, rather than high instantaneous power.
The lifespan of a diamond battery is determined by the half-life of the radioactive isotope used. For instance, if Carbon-14 is used (which has a half-life of 5,730 years), the battery could effectively produce power for thousands of years, far exceeding conventional batteries.
No, diamond batteries are not designed to replace traditional fossil fuels or large-scale grid power. Their low power output makes them unsuitable for immediate high-energy demands. Instead, they are suited for specialized, long-term, low-power applications where reliability and longevity are paramount.
Common radioactive isotopes being researched for diamond batteries include Carbon-14 (C-14), which is a byproduct of nuclear reactor operations, and Nickel-63 (Ni-63) or Tritium (H-3) due to their suitable beta decay properties.
Verified Expert
Alex Rivers
A professional researcher since age twelve, I delve into mysteries and ignite curiosity by presenting an array of compelling possibilities. I will heighten your curiosity, but by the end, you will possess profound knowledge.
Leave a Reply
Comments (0)
No approved comments yet. Be the first to share your thoughts!
Leave a Reply
Comments (0)