I remember staring up at the night sky as a child, utterly captivated by the seemingly endless sprawl of stars. Every speck of light felt like a story, a mystery waiting to be unravelled. It’s a feeling that has never truly left me. More than just pretty lights, those celestial bodies, and the vast, dark spaces between them, hold the secrets to our very existence. One of the most profound questions we can ask is: **where did life come from?** For decades, scientists have pursued this inquiry, often looking at early Earth as the crucible where the first organic molecules, the building blocks of life, were forged. But what if the origin story is far more cosmic, more ancient, and far more pervasive? What if the seemingly inert dust scattered across the universe isn't just cosmic debris, but rather **tiny factories diligently assembling the very components that make life possible?**
This isn't science fiction; it's a rapidly evolving field of astrochemistry that suggests the interstellar medium — the gas and dust residing between stars — is far from empty. In fact, it might be a vibrant, albeit cold and diffuse, chemical laboratory. The idea that life’s essential ingredients could be cooked up in the freezing vacuum of space challenges our traditional, Earth-centric view of abiogenesis. It opens up a universe of possibilities, where the seeds of life might be universal, waiting to germinate wherever conditions allow.
The Cosmic Ingredient List: More Than Just Dust
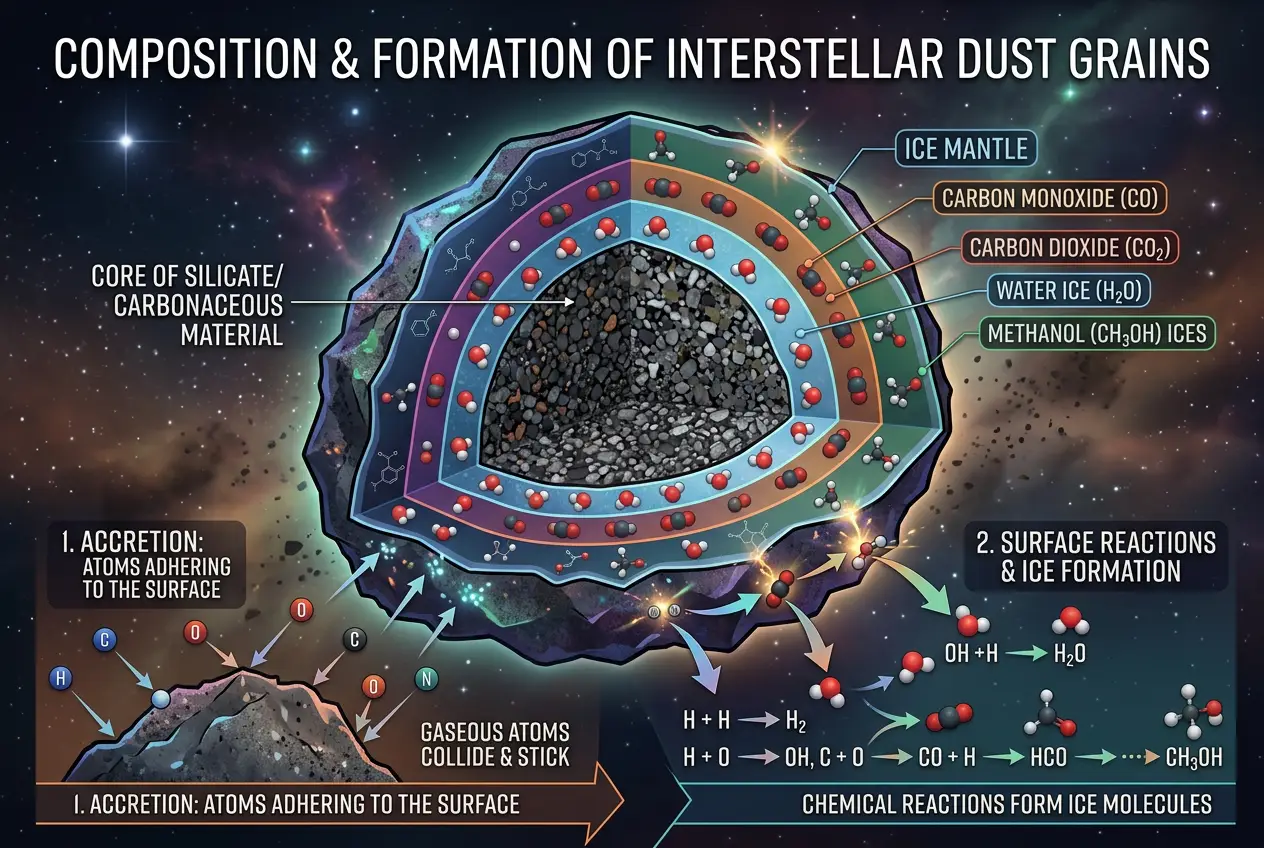
When we talk about "space dust," we're not talking about household lint. Instead, imagine microscopic particles, often no larger than a smoke particle, composed primarily of **silicates** (rock-like materials) and **carbonaceous matter** (soot-like materials), often coated with various ice mantles of water, carbon monoxide, carbon dioxide, and methanol. These tiny grains are suspended in the **interstellar medium (ISM)**, a vast expanse of gas and dust that fills the space between star systems within a galaxy. The ISM is where stars are born, and it’s a dynamic environment, constantly recycled through stellar birth and death. For a deeper dive into the basic components of the cosmos, you might find our previous discussion on
the interstellar medium on Wikipedia quite illuminating.
What makes these dust grains so special? Their surfaces. While the ISM is incredibly diffuse, meaning atoms and molecules rarely collide in the gas phase, the surfaces of these dust grains provide a crucial meeting point. Think of them as **catalytic platforms** – miniature reaction chambers where atoms and simple molecules can stick, accumulate, and react with each other under specific conditions.
Tiny Factories in the Void: How Chemistry Happens in the Cold
The conditions in interstellar space are extreme. Temperatures can plunge to just a few degrees above absolute zero (around -263°C), and the vacuum is far more profound than anything achievable on Earth. Yet, it’s precisely these conditions that, counter-intuitively, facilitate the formation of complex molecules.
Here's a simplified breakdown of the process:
1. **Adsorption:** Atoms and simple molecules (like hydrogen, oxygen, carbon, nitrogen) float through the ISM. When they encounter a dust grain, they can stick to its surface. This process is called **adsorption**.
2. **Migration:** Once adsorbed, these atoms and molecules aren't necessarily stuck in one spot. They can slowly migrate across the grain's surface, driven by tiny thermal energies or quantum tunneling.
3. **Reaction:** When two or more reactive species encounter each other on the surface, they can react to form a new, more complex molecule. The dust grain effectively lowers the energy barrier for these reactions.
4. **Desorption:** Once a new molecule is formed, it can either remain on the grain (often getting incorporated into the ice mantle) or be released back into the gas phase, perhaps by the energy from a nearby forming star, UV radiation, or even cosmic rays.
This "surface chemistry" is remarkably efficient. Without the dust grains, the likelihood of two specific atoms colliding in the vast emptiness of space and reacting is astronomically low. The grains concentrate the reactants, making chemical synthesis possible even in the most hostile environments.
The Chemistry of the Cold: When Ice Becomes a Catalyst
The ice mantles covering many dust grains play an even more critical role. These icy layers aren't just frozen water; they're mixtures of various molecules that can act as solvents or even participate in reactions themselves. Imagine a miniature, frozen reaction flask. As UV radiation from nearby stars or cosmic rays penetrates these ice mantles, it can break chemical bonds, creating highly reactive free radicals. These radicals then recombine to form increasingly complex molecules. This process, known as **photochemistry** and **radiolysis** within the ice, is a powerful engine for building organic complexity.
I've learned that one of the fascinating aspects of this is how molecules like formaldehyde (H2CO), methanol (CH3OH), and even more complex species like glycolaldehyde (a simple sugar) can form under these conditions. These aren't just random chemicals; they are direct precursors to many of life's fundamental components.
Complex Molecules Emerge: The Universe's Hidden Pantry
Astronomers, using powerful telescopes like the Atacama Large Millimeter/submillimeter Array (ALMA) and the James Webb Space Telescope (JWST), have been discovering an astonishing array of complex organic molecules in interstellar clouds, protoplanetary disks, and even comets. These include:
* **Amino Acids:** The building blocks of proteins. While whole proteins haven't been found, their precursor amino acids have.
* **Sugars:** Simple sugars like glycolaldehyde have been detected. These are crucial components of RNA and DNA.
* **Nucleobases:** The fundamental units of genetic material (adenine, guanine, cytosine, thymine/uracil). While direct detections are challenging, chemical pathways for their formation in space are being investigated.
* **Polycyclic Aromatic Hydrocarbons (PAHs):** Large carbon-based molecules that are ubiquitous in space and are thought to be important for chemical evolution.
These discoveries provide compelling evidence that the universe is rich with the raw materials for life. The field dedicated to understanding these cosmic chemical reactions is called
astrochemistry, as explored on Wikipedia.
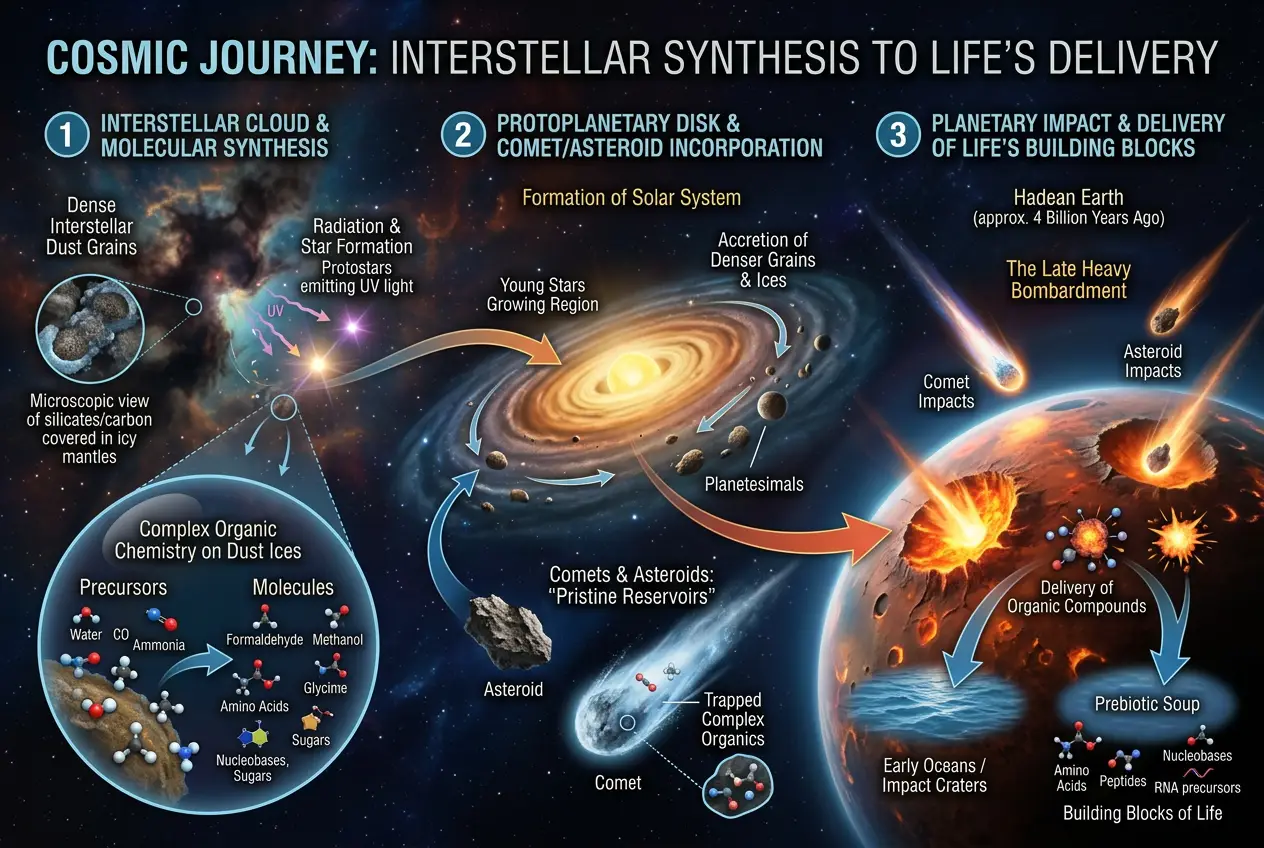
From Dust to Planets to Life: A Cosmic Delivery Service
So, if complex organic molecules are forming in space dust, how do they get to planets? This is where comets and asteroids come in. As new solar systems form, the dust and gas in the protoplanetary disk coalesce into planets, asteroids, and comets. These icy and rocky bodies can effectively **trap and preserve** the organic molecules formed in the earlier interstellar phase.
When comets and asteroids impact young planets, they act as cosmic delivery trucks, depositing these pre-formed organic molecules onto the planetary surface. This mechanism aligns with theories like **panspermia**, which suggests life's ingredients (or even life itself) could have been seeded from space. If you're curious about how celestial bodies might have seeded life, check out our blog post on
Could Meteors Seed Life? The Panspermia Puzzle. While panspermia often discusses living organisms, the delivery of pre-biotic molecules is a crucial, less speculative aspect.
Lab Experiments & Observational Evidence: Replicating the Cosmos
Scientists aren't just theorizing; they're actively experimenting. **Astrophysical simulation chambers** on Earth replicate the extreme cold and vacuum of space, allowing researchers to observe how organic molecules form on simulated dust grain surfaces under various conditions. These experiments have successfully demonstrated the formation of many of the complex molecules detected by telescopes.
Moreover, the increasing resolution and sensitivity of telescopes continue to provide more and more direct evidence. I find it absolutely astounding that we can peer across vast cosmic distances and detect the spectral signatures of molecules that are literally the foundation of life. Each new detection reinforces the idea that the universe is inherently pre-programmed, in a chemical sense, to produce life's building blocks. It makes you wonder about the broader implications for the universe itself, perhaps even its fundamental nature, as discussed in topics like
Does Dark Matter Hide a Universe We Can't See?, where hidden components influence everything.
Implications for Life Beyond Earth: A Universe Teeming with Potential?
The profound implication of space dust acting as cosmic molecular factories is that **the building blocks of life might be common throughout the universe.** If these complex organic molecules can form spontaneously and efficiently in diverse interstellar environments, then every star system with planets could potentially receive a generous supply of these essential ingredients.
This significantly boosts the probability of life arising elsewhere. Instead of life being a freak accident specific to Earth's early conditions, it suggests a universal biochemical tendency. It pushes the question from "How did life start?" to "How often does it *fail* to start, given the ubiquity of its components?" This perspective also ties into the concept that life might leverage fundamental cosmic principles, a fascinating idea when considering concepts like
Is Human DNA a Cosmic Antenna?, hinting at deeper connections between life and the cosmos.
Challenges and Future Research
Despite these exciting advancements, many questions remain. What is the precise pathway for the formation of the most complex molecules, like nucleobases, in interstellar space? How efficiently do these molecules survive the harsh journey from interstellar cloud to planetary surface? What specific environmental conditions are most conducive to their formation and delivery?
Future missions, both orbital telescopes and sample return missions from comets and asteroids, will undoubtedly provide more clues. Understanding the exact chemical inventory delivered to early Earth is crucial for piecing together the complete picture of abiogenesis.
Conclusion: The Universe's Intricate Dance
The journey to understand life’s origins is one of the most compelling scientific endeavors. While the notion that Earth was the sole architect of life's initial components is a comforting thought, the evidence increasingly points to a grander, more cosmic narrative. **Space dust, those seemingly insignificant specks in the void, might be orchestrating an intricate chemical dance, patiently assembling the very first steps towards life.** It’s a testament to the universe’s incredible capacity for complexity and a powerful reminder that the wonders of the cosmos are often found in the most unexpected, and seemingly mundane, places. The next time I gaze at the Milky Way, I’ll see not just stars, but billions of tiny chemical workshops, tirelessly working to craft the ingredients for life itself.

 Join Us
Join Us







































Leave a Reply
Comments (0)