I often find myself contemplating the unseen forces that shape our existence – the invisible currents that power our homes, the silent waves that carry our voices, the very fabric of reality that we so often take for granted. It’s a remarkable thought, isn't it? That much of what we experience daily is built upon principles discovered by minds centuries ago, grappling with mysteries that seemed almost magical. Today, I want to invite you on an extraordinary journey, a deep dive into the life of one such mind: Michael Faraday.
This will be a very long and comprehensive blog post, meticulously detailing the life, struggles, and monumental discoveries of a man who, despite humble beginnings, fundamentally reshaped our understanding of electricity, magnetism, and the very nature of physical reality. His story isn't just a testament to scientific genius, but to perseverance, self-education, and an unwavering commitment to unraveling the universe’s secrets.
### From Apprentice to Alchemist of the Invisible: The Unlikely Start
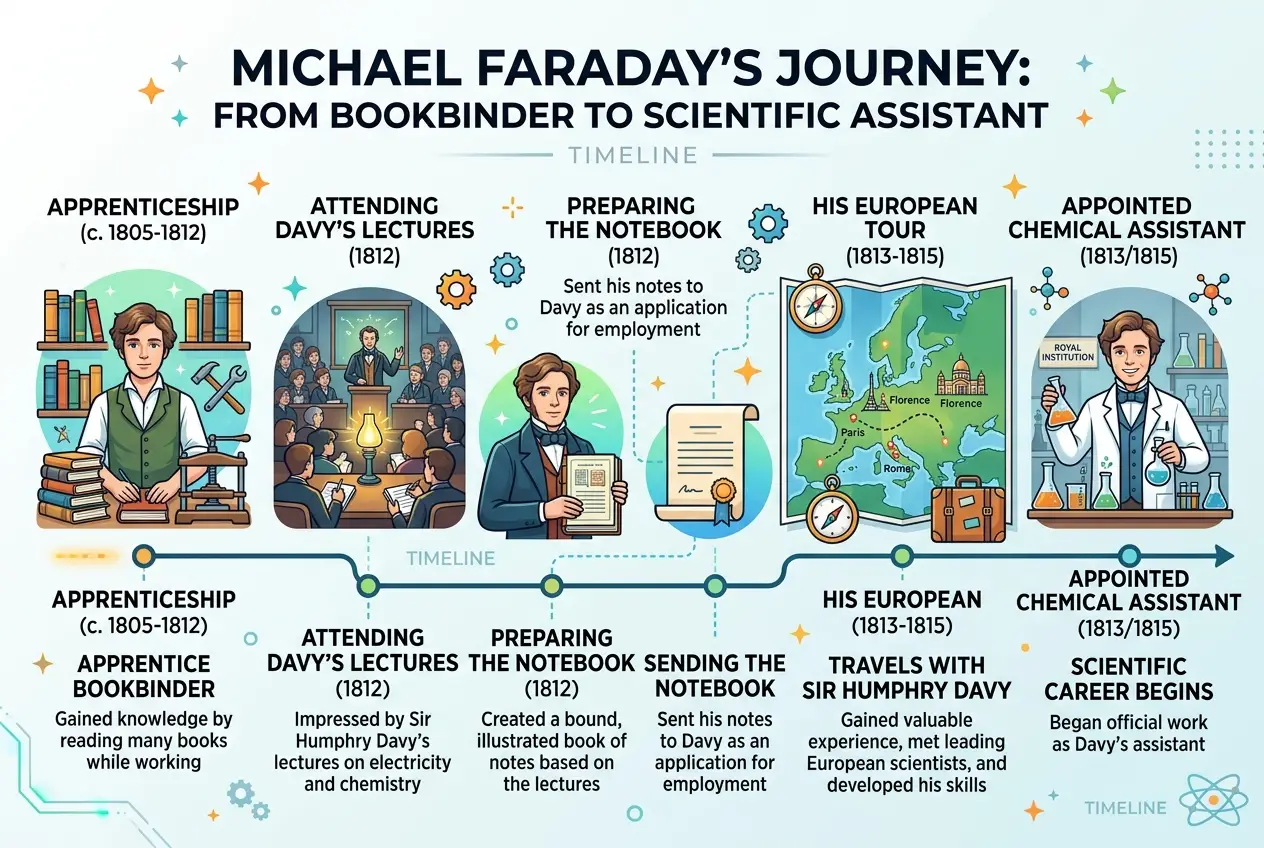
Imagine a young boy in 18th-century London, barely able to afford an education, his hands stained with printer's ink rather than laboratory chemicals. This was Michael Faraday, born in 1791 in Newington Butts, just south of the River Thames. His father, James Faraday, was a blacksmith suffering from ill health, and his mother, Margaret, struggled to keep their family afloat amidst grinding poverty. Formal schooling was a luxury they couldn't afford, so young Michael's education was minimal, focusing primarily on reading, writing, and arithmetic – just enough to get by.
At the tender age of 14, Faraday’s path took a crucial turn. He was apprenticed to George Riebau, a local bookbinder and stationer. For seven years, he meticulously bound books, repaired pages, and handled countless volumes that passed through his hands. This seemingly mundane occupation, however, became his university. Surrounded by knowledge, Faraday devoured every science book he could lay his hands on. He was particularly captivated by conversations on chemistry and electricity, reading articles in the *Encyclopaedia Britannica* and immersing himself in Jane Marcet's *Conversations on Chemistry*, which explained complex scientific principles in an accessible dialogue format. This period of self-study wasn't just about absorbing information; it ignited a spark of profound curiosity that would define his entire life.
I sometimes wonder what it must have felt like for him, poring over those texts late into the night, dreaming of experiments he could only read about. It wasn't merely rote learning; it was a deeply personal quest for understanding. He would save the few pennies he earned to buy simple equipment, conducting rudimentary experiments in his small room, fueled by an insatiable desire to see these principles in action.
### The Golden Ticket: A Chance Encounter with Humphry Davy
The turning point in Faraday’s life arrived in 1812. A customer at Riebau's shop, a man named William Dance, noticed Faraday’s enthusiasm for science and offered him tickets to attend four lectures by the celebrated chemist Sir Humphry Davy at the Royal Institution. Davy was a rock star of his time, famous for isolating elements like sodium and potassium, and for his dramatic and engaging lecture style.
For Faraday, these lectures were nothing short of a revelation. He meticulously took notes, illustrating Davy's experiments and later neatly binding them into a 386-page book. This volume, a testament to his diligence and scientific aptitude, became his "golden ticket." When Davy temporarily damaged his eyesight in an explosion, Faraday seized the opportunity. He sent his bound lecture notes, along with a polite letter expressing his desire to work in science, to Davy.
Davy, initially hesitant due to Faraday's humble background and lack of formal education, was nevertheless impressed by the young man's initiative and meticulous record-keeping. As he famously quipped, "I confess I saw in him a vivacity of eye, a quickness of apprehension, a promptness of memory, a mildness of temper, and an obliging disposition, which made me desire to retain him." In March 1813, Faraday was offered a position as a chemical assistant at the Royal Institution, marking his official entry into the world of professional science.
This was a profound moment, not just for Faraday, but for the trajectory of scientific discovery. I often reflect on how serendipitous this encounter was. Had Dance not offered those tickets, had Faraday not been so diligent, had Davy not seen beyond social class, the world might have waited decades longer for the marvels Faraday would unlock.
### A Grand Tour and Gaining Experience (1813-1815)
Shortly after joining the Royal Institution, Faraday embarked on a grand tour of Europe as Davy’s scientific assistant and valet. This journey, lasting nearly two years, took them across France, Italy, and Switzerland, introducing Faraday to some of the greatest scientific minds of the era, including André-Marie Ampère in Paris and Alessandro Volta in Milan. For a young man who had never left London, this was an unparalleled educational experience, immersing him in the scientific discourse and laboratory practices of the continent’s leading researchers.
However, the tour was not without its challenges. Davy's wife, Jane Apreece, reportedly treated Faraday more as a servant than a scientific colleague, often making him eat with the servants and engaging in petty slights. Despite these social humiliations, Faraday remained focused, absorbing every detail of the scientific work conducted in the European laboratories and expanding his practical skills. He learned invaluable laboratory techniques and began to understand the nuances of experimental design. This period was crucial for his development, solidifying his hands-on expertise and sharpening his observational skills.
### The Independent Chemist: Early Triumphs (1816-1825)
Upon returning to the Royal Institution, Faraday's career as an independent researcher began to blossom. Though still assisting Davy, he started publishing his own scientific papers, initially focusing on analytical chemistry. His meticulous approach and keen observational skills quickly made him adept at chemical analysis. He published over 40 papers on various chemical topics, contributing significantly to the understanding of chlorine, nitrogen, and carbon compounds.
One of his most notable early achievements was the **liquefaction of gases**. Scientists at the time believed that gases were permanently elastic fluids. In 1823, Faraday, with Davy’s encouragement, successfully liquefied chlorine gas by cooling and compressing it. He later achieved the same with other gases, including carbon dioxide and sulfur dioxide. This groundbreaking work demonstrated that gases were simply vapours of easily evaporable liquids and helped establish the concept of a critical temperature, beyond which a gas cannot be liquefied, regardless of pressure. You can explore more about this concept on Wikipedia's page on [liquefaction of gases](https://en.wikipedia.org/wiki/Liquefaction_of_gases).
Another significant discovery came in 1825: **Benzene**. While investigating oily residues in gas cylinders used for illuminating London, Faraday isolated a new compound, a cyclic hydrocarbon he named bicarburet of hydrogen. We now know it as benzene, a fundamental building block in organic chemistry. This discovery had profound implications for industrial chemistry and paved the way for the vast field of aromatic chemistry.
By this time, Faraday’s reputation was growing. He was promoted to Superintendent of the House at the Royal Institution and then, in 1825, became the Director of the Royal Institution’s Laboratory, succeeding Davy. His personal life also found stability with his marriage to Sarah Barnard in 1821, a union that provided him with immense emotional support throughout his life.
### The Spark of Electromagnetism: A Revolution Begins (1820-1831)
The 1820s proved to be the decade where Faraday truly began to shine, shifting his focus towards the burgeoning field of electricity and magnetism. The scientific community was abuzz with Hans Christian Ørsted’s 1820 discovery that an electric current creates a magnetic field. This revelation was a game-changer, demonstrating a fundamental connection between two previously separate forces.
Many scientists, including Humphry Davy and William Hyde Wollaston, attempted to build upon Ørsted’s work, trying to make a wire carrying current rotate around a magnet or a magnet around a wire. They envisioned a continuous motion, but their efforts were unsuccessful.
Faraday, with his intuitive understanding of forces, approached the problem differently. In 1821, he designed an ingenious experiment. He suspended a wire carrying current into a pool of mercury, with a magnet placed vertically in the centre. When the current flowed, the wire continuously rotated around the magnet. He then inverted the setup, allowing a magnet to rotate around a fixed wire. These were the first continuous **electric motors**, a monumental invention that laid the groundwork for all future electromechanical devices.
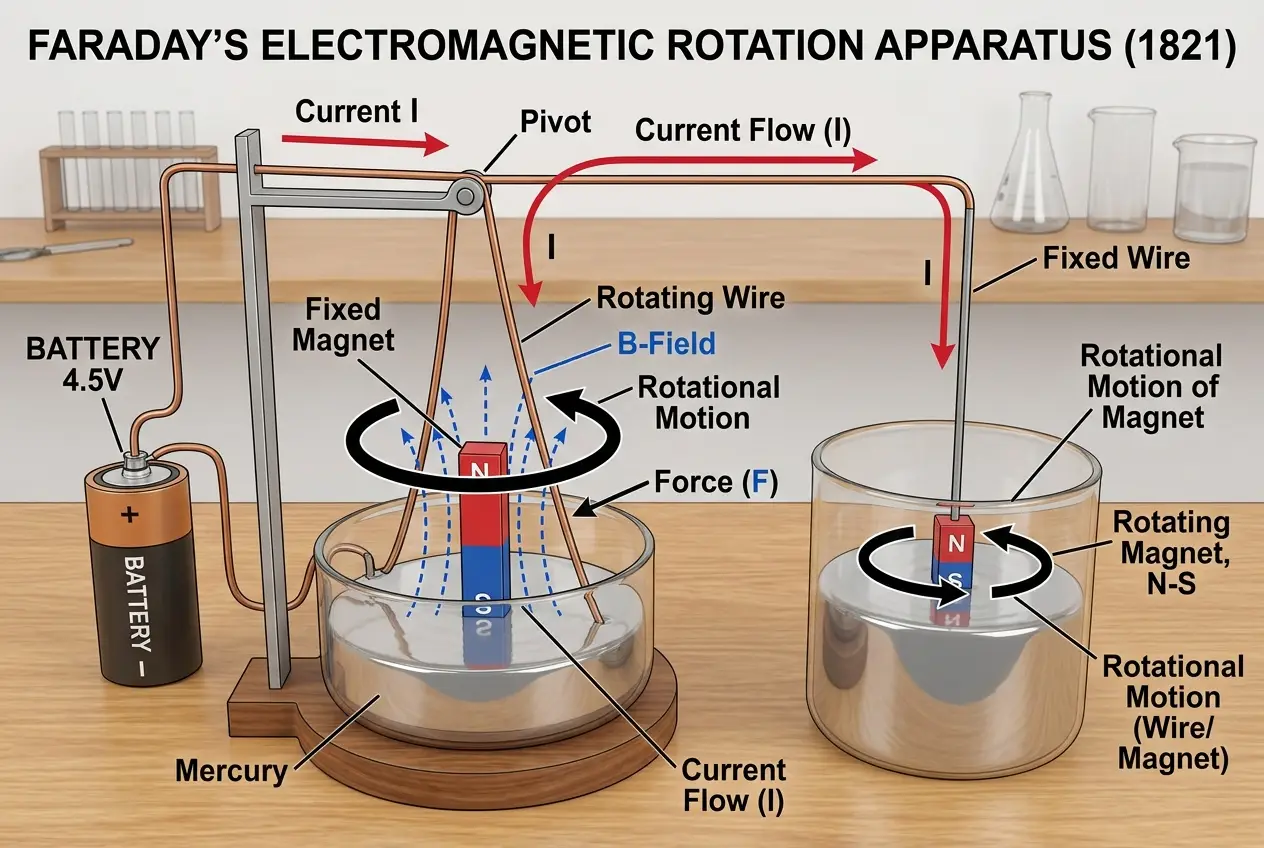
This breakthrough, however, led to an unfortunate controversy. Faraday published his results without adequately crediting Wollaston, who had reportedly discussed similar ideas with Davy. Though Faraday later apologized and clarified his position, the incident created a rift with some members of the scientific establishment, including his mentor, Davy. Despite this, his election to the Royal Society in 1824 was a clear acknowledgment of his genius, even as Davy reportedly opposed it.
### The Magnum Opus: Electromagnetic Induction (1831)
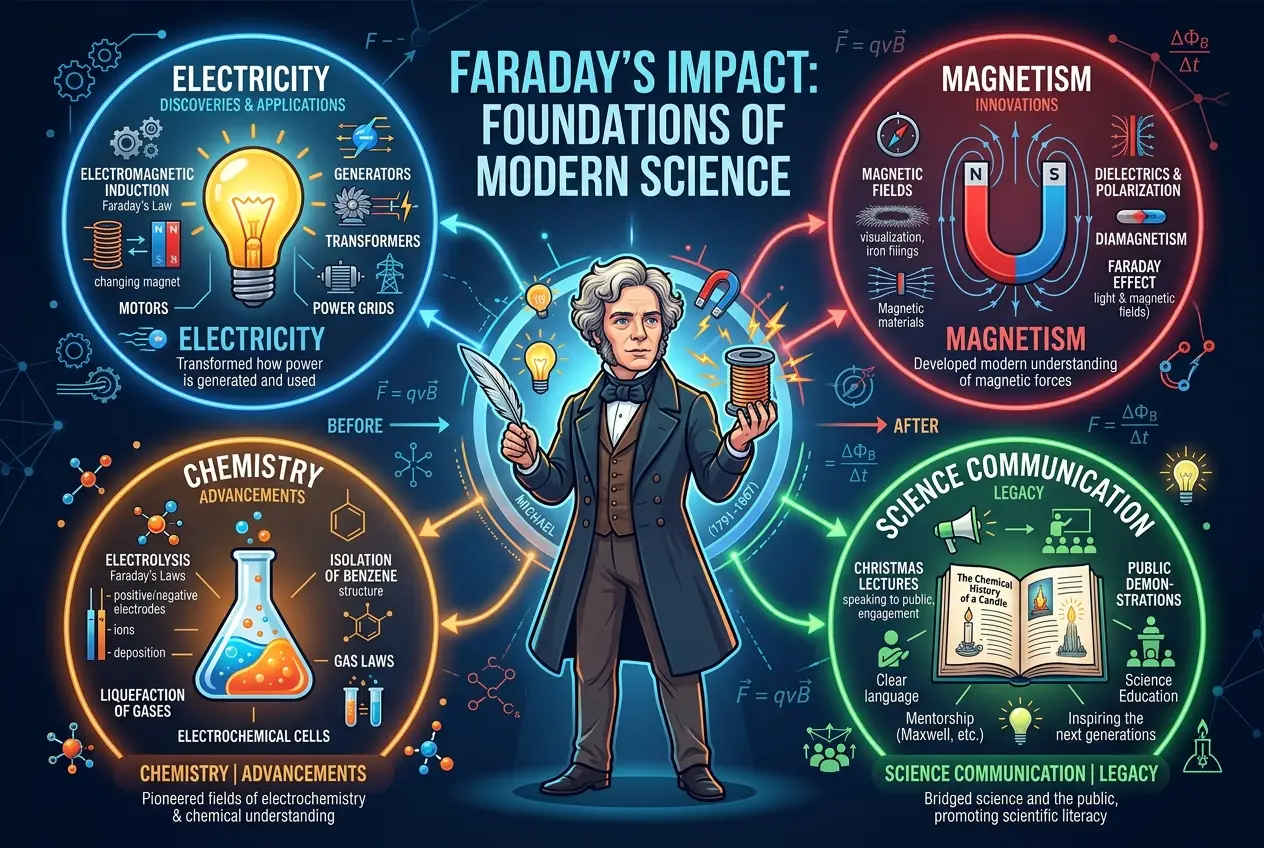
If the electric motor was a flash of brilliance, Faraday’s discovery of **electromagnetic induction** was a sustained burst of revolutionary insight that would forever change the world. For years, he had pondered the inverse of Ørsted’s discovery: if electricity could produce magnetism, could magnetism produce electricity?
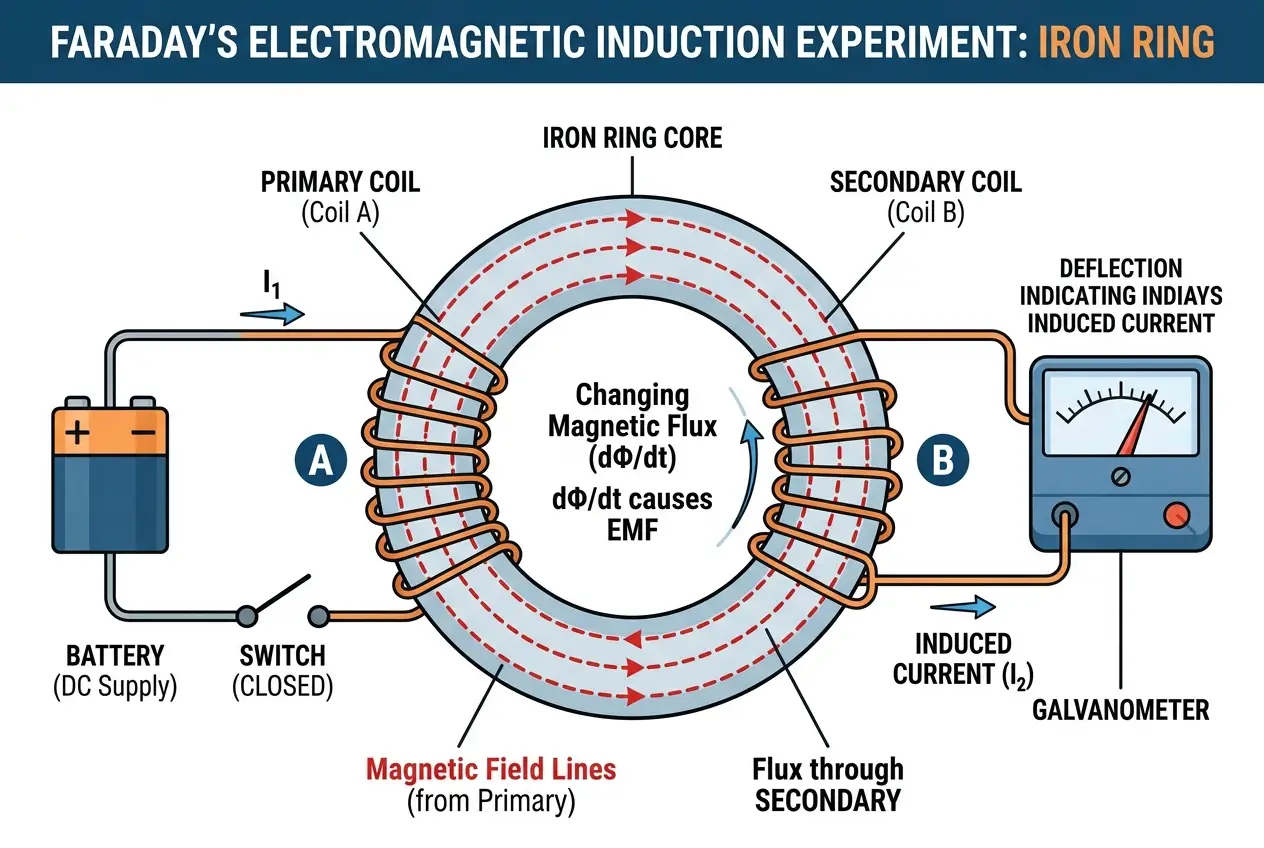
In August 1831, Faraday began a series of meticulous experiments. He wound two separate coils of insulated copper wire around opposite sides of a soft iron ring, which served as a core. One coil was connected to a battery (the primary circuit), and the other to a galvanometer (to detect current, the secondary circuit). The moment he connected or disconnected the primary circuit to the battery, the galvanometer in the secondary circuit would show a momentary deflection, indicating the presence of an induced current. While the primary current was steady, there was no induction. This meant that it wasn't the steady magnetic field that produced electricity, but the *change* in the magnetic field.
This was an incredible realization. He further demonstrated this by moving a magnet in and out of a coil of wire. Again, a current was induced, but only when the magnet was in motion. The faster the motion, the greater the induced current. This elegantly simple experiment proved that a changing magnetic field induces an electric current. This is now known as **Faraday's Law of Induction**, a cornerstone of electromagnetism. You can read more about it here: [Faraday's Law of Induction on Wikipedia](https://en.wikipedia.org/wiki/Faraday%27s_law_of_induction).
This discovery was the genesis of the **electric generator** and the **transformer**. It meant that mechanical energy could be converted into electrical energy, paving the way for large-scale power generation. It was the crucial step towards harnessing electricity for practical use, moving beyond chemical batteries and static electricity. Our entire modern electrical infrastructure, from power plants to phone chargers, owes its existence to this singular insight. This was a direct progression from the initial steps in understanding electricity, some of which were seen in the intriguing "Baghdad Battery" that raises questions about whether ancient civilizations harnessed electricity. You can explore more about that historical tech at [The Baghdad Battery: Could Ancient Civilizations Harness Electricity?](blogs/baghdad-battery-did-ancients-spark-our-electric-age-7832).
### Unifying Forces: Electrochemistry and the Field Concept (1832-1839)
Faraday was not content merely with discovery; he sought to understand the underlying principles and connections between different phenomena. His work on electromagnetic induction led him to investigate **electrochemistry** in greater depth. He wanted to understand how electricity interacted with chemical compounds, particularly during electrolysis, the process of using electricity to drive non-spontaneous chemical reactions.
Between 1832 and 1834, he formulated his **Laws of Electrolysis**, which precisely quantified the relationship between the amount of electricity passed through an electrolyte and the amount of substance deposited or liberated at the electrodes. He introduced terms that are still fundamental to chemistry today: **electrode**, **anode**, **cathode**, **ion**, and **electrolyte**. His research demonstrated that the amount of chemical change produced by an electric current is proportional to the quantity of electricity that passes, and that the mass of a substance deposited or dissolved is proportional to its electrochemical equivalent.
Crucially, Faraday's electrochemical studies led him to propose the revolutionary idea of the **unity of electrical forces**. At the time, various forms of electricity were thought to be distinct: static electricity from friction, voltaic electricity from batteries, animal electricity from living creatures. Faraday painstakingly demonstrated that all these forms were manifestations of the same fundamental force, differing only in intensity and quantity. This was a profound step towards a unified field theory, hinting at a universe governed by interconnected principles rather than disparate forces. This foundational understanding laid important groundwork for later thinkers like Nikola Tesla, another enigmatic genius who wired our modern world. You can learn more about him in our blog, [Nikola Tesla: The Enigmatic Genius Who Wired Our Modern World](blogs/nikola-tesla-the-enigmatic-genius-who-wired-our-modern-world-2839).
His vision was that of **fields of force** permeating space, rather than action at a distance. He imagined lines of electric and magnetic force extending from charged bodies and magnets, influencing objects within their reach. This departure from Newton's instantaneous action at a distance was radical for its time but would become central to modern physics. He spent years visualizing these invisible fields, often sketching them, trying to make tangible what no one could see. This intuitive grasp of fields was perhaps his greatest conceptual contribution.
### Further Explorations: Light, Magnetism, and Decline (1840-1867)
The sheer intensity of Faraday’s work began to take a toll on his health. In the early 1840s, he suffered from a period of severe nervous fatigue and memory loss, which forced him to take a break from active research. When he returned, his focus remained on his grand vision of unifying the forces of nature, particularly the connection between light, electricity, and magnetism.
In 1845, he made another monumental discovery: the **magneto-optical effect**, also known as the **Faraday effect**. He found that when linearly polarized light passes through a transparent dielectric material in the presence of a strong magnetic field aligned with the light's direction of propagation, the plane of polarization of the light rotates. This was the first experimental evidence of a direct relationship between light and magnetism, paving the way for the understanding that light itself is an electromagnetic phenomenon. It was a crucial precursor to James Clerk Maxwell’s theory of electromagnetism. Faraday’s work effectively showed that a cosmic computer, or at least a highly interconnected universe, was at play. This resonates with the modern question: [Is the Universe a Giant Neural Network?](blogs/is-the-universe-a-giant-neural-network-2907).
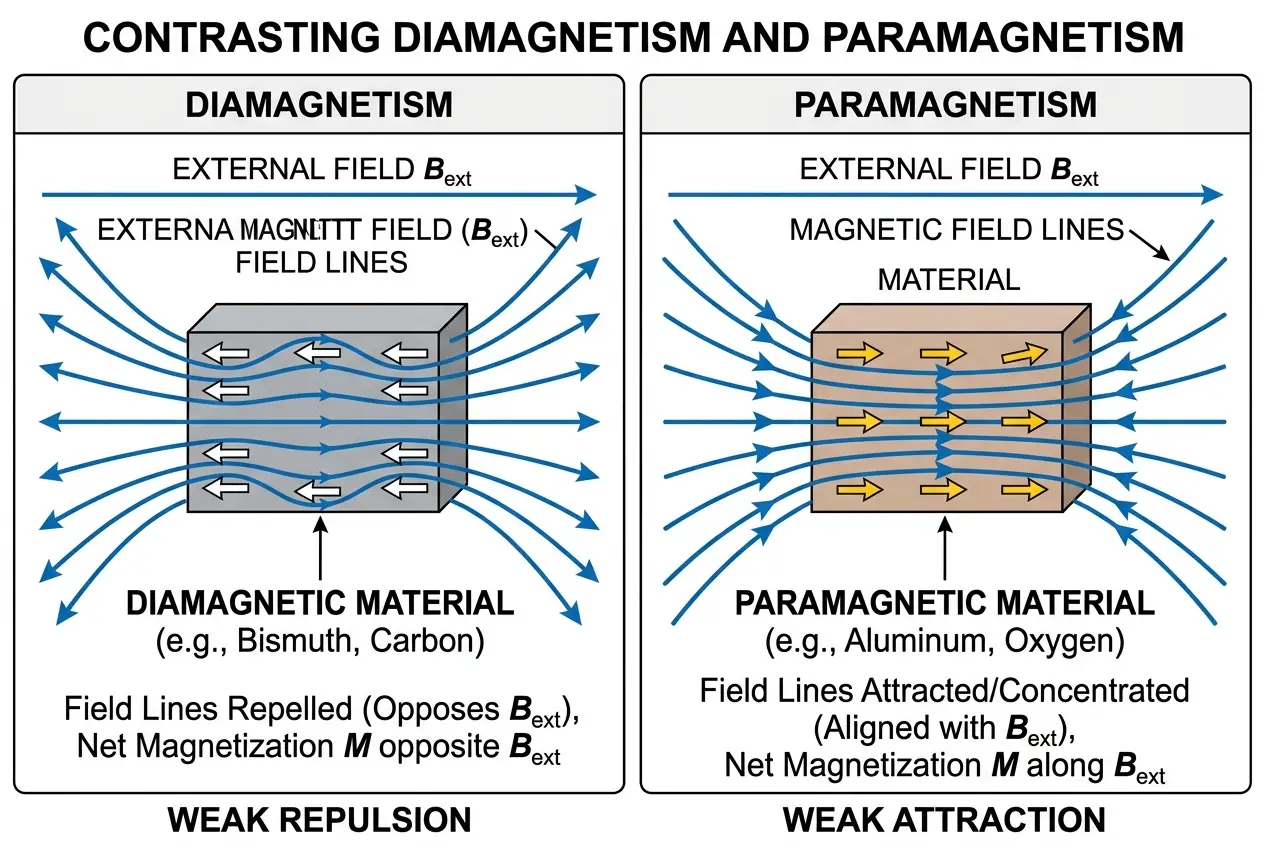
In the same year, he discovered **diamagnetism** and **paramagnetism**. He observed that all substances are either attracted or repelled by a magnetic field, classifying them based on their interaction. Diamagnetic materials, like bismuth or water, are weakly repelled, while paramagnetic materials, like aluminum or oxygen, are weakly attracted. This further cemented his belief in the universal influence of magnetic forces.
Throughout these later years, Faraday continued to develop his concept of **lines of force**, emphasizing their physical reality. He saw them not as mere mathematical constructs but as dynamic entities that filled space, mediating interactions between bodies. This highly intuitive and visual approach profoundly influenced a young Scottish physicist named James Clerk Maxwell, who would later translate Faraday’s qualitative field concepts into a precise mathematical framework, giving us Maxwell's Equations – the foundation of classical electromagnetism. These equations are as fundamental as Einstein’s relativity in describing reality.
Despite his immense contributions, Faraday remained a humble and principled man. He famously refused a knighthood and the presidency of the Royal Society, preferring to remain "plain Mr. Faraday," dedicated solely to his scientific pursuits and public service. His integrity and commitment to science above personal accolades set a powerful example.
### Public Service and the Popularization of Science
Faraday was not just a groundbreaking researcher; he was a passionate advocate for science education and public welfare. He initiated the **Christmas Lectures for Children** at the Royal Institution in 1825, a tradition that continues to this day. These lectures, delivered with captivating demonstrations and engaging explanations, aimed to ignite scientific curiosity in young minds. His most famous lecture series, "The Chemical History of a Candle," is a masterpiece of accessible science communication, explaining fundamental scientific principles through the simple act of burning a candle.
Beyond the lecture hall, Faraday applied his scientific expertise to practical problems. He worked extensively on improving the lighting in **lighthouses**, advocating for more efficient and powerful lamps to prevent shipwrecks. His expertise was sought for matters of public health, such as investigating air pollution in London and the infamous **Great Stink of 1858**, where the stench from the River Thames became unbearable. He advised on sanitation and ventilation, demonstrating a keen sense of social responsibility. His dedication to clear public communication about science is a reminder of its importance, even in today's digital age. We often delve into how complex scientific concepts impact daily life, such as in our blog on [Why is Quantum Computer 1 Million Times Faster Than SuperComputers?](blogs/why-is-quantum-computer-1-million-times-faster-than-supercomputers-).
### A Quiet End and an Enduring Legacy
As he entered his final years, Faraday's health continued to decline, marked by increasing memory loss and weakness. Queen Victoria, in recognition of his immense contributions to science, provided him with a grace and favour house at Hampton Court, where he spent his last years in relative tranquility. He passed away peacefully on August 25, 1867, at the age of 75.
His death, though marking the end of a remarkable life, was merely the beginning of his enduring legacy. Michael Faraday left behind a world utterly transformed by his discoveries.
### His Legacy Can Be Summarized In Several Key Areas:
* **The Foundation of Electrical Engineering:** Every electric generator, every transformer, every electric motor traces its lineage back to Faraday’s initial experiments on electromagnetic rotation and induction. His work paved the way for industrial electricity, making possible the vast power grids and electronic devices we rely on today. Without Faraday, the second industrial revolution would have been inconceivable.
* **The Concept of the Field:** Faraday’s intuitive and persistent development of the "field" concept, where forces are mediated through space rather than acting instantaneously at a distance, revolutionized physics. It moved away from mechanical models and introduced a profound new way of understanding interactions. This concept was fundamental for Maxwell's mathematical formulation of electromagnetism and later for Einstein's theories of relativity, where spacetime itself became a field. His ideas of invisible forces resonating through space even echo in modern theories about the universe's structure, like the cosmic web. You can read more about it here: [The Cosmic Web: Does it Compute Our Universe’s Fate?](blogs/can-the-cosmic-web-compute-our-universes-fate-1603).
* **Unifying Forces:** His demonstration of the unity of various forms of electricity and the connection between light and magnetism laid crucial groundwork for the ultimate goal of physics: a unified theory of everything. He showed that seemingly disparate phenomena were deeply interconnected, driven by a few fundamental forces.
* **Pioneer of Science Communication:** His Christmas Lectures and public demonstrations made science accessible and exciting for generations. He believed deeply that scientific knowledge should not be confined to academic circles but shared with the broader public.
* **A Model of Scientific Method:** Faraday’s meticulous experimental technique, his insistence on direct observation, and his relentless pursuit of understanding phenomena, rather than just describing them, set a gold standard for scientific inquiry. He worked with simple, often homemade, apparatus, proving that profound discoveries could emerge from keen observation and careful experimentation, not just expensive equipment.
* **Benzene and Organic Chemistry:** His isolation of benzene was a significant contribution to chemistry, opening up a vast field of research into organic compounds and their structures, which underpin countless modern industries, from pharmaceuticals to plastics.
Michael Faraday was a giant of science, a self-made genius who saw deeper into the fabric of nature than perhaps anyone before him. His mind, unburdened by conventional academic dogma, approached problems with a fresh perspective, guided by intuition and an insatiable desire for truth. He gave us the means to light our cities, power our machines, and understand the very nature of light itself.
I believe his story serves as a profound reminder that curiosity, perseverance, and a deep sense of wonder are the true engines of progress. He started with nothing but an inquisitive mind and a stack of books, and he ended up illuminating the entire modern world, quite literally. His legacy is not just a collection of discoveries, but an enduring inspiration to all who dare to look beyond the obvious and seek the hidden mechanisms of our universe. What hidden forces, I wonder, are waiting for our own generation to uncover?
Blog Idea By Venkat Faraday ( Source: Comment On Pinterest )
Frequently Asked Questions
Michael Faraday's journey was marked by extraordinary self-education and an insatiable curiosity. As a bookbinder's apprentice, he devoured scientific texts, fueling his passion. A serendipitous encounter led him to become an assistant to Sir Humphry Davy at the Royal Institution, where his meticulous work ethic and brilliant intuition propelled him to scientific stardom, despite a lack of formal schooling.
Faraday's most significant contributions include the discovery of electromagnetic induction (the principle behind electric generators and transformers), the invention of the first electric motor, the formulation of the Laws of Electrolysis, the discovery of benzene, and the establishment of the concept of the electromagnetic field. He also discovered the magneto-optical effect, linking light and magnetism.
Faraday's qualitative concept of 'lines of force' and the electromagnetic field profoundly influenced James Clerk Maxwell. Maxwell translated Faraday's intuitive ideas into a precise set of mathematical equations, known as Maxwell's Equations, which are the foundation of classical electromagnetism. These equations, in turn, were crucial for Albert Einstein's development of the theory of special relativity, as they demonstrated the constant speed of light and the interconnectedness of electric and magnetic fields.
Faraday was a passionate advocate for making science accessible to everyone. He initiated the famous Christmas Lectures for Children at the Royal Institution, which continue to this day, and delivered captivating public demonstrations. His lecture series, 'The Chemical History of a Candle,' is still celebrated as a model for explaining complex scientific principles in an engaging and understandable way.
Faraday was a devout member of the Sandemanian Christian sect, which emphasized humility, integrity, and direct interpretation of the Bible. His faith deeply influenced his scientific work, fostering a belief in a coherent, divinely ordered universe governed by unified laws. This spiritual outlook encouraged his relentless search for connections between seemingly disparate phenomena and his commitment to truth over personal gain, exemplified by his refusal of a knighthood.
Before Faraday, forces like gravity or magnetism were often explained as 'action at a distance.' Faraday, however, proposed that these forces were mediated by invisible 'fields' permeating space, with 'lines of force' extending from objects. This revolutionary concept allowed physicists to visualize and mathematically describe how forces interact locally, profoundly changing the understanding of fundamental interactions and laying the groundwork for modern field theories in physics.
Verified Expert
Alex Rivers
A professional researcher since age twelve, I delve into mysteries and ignite curiosity by presenting an array of compelling possibilities. I will heighten your curiosity, but by the end, you will possess profound knowledge.
Leave a Reply
Comments (0)
No approved comments yet. Be the first to share your thoughts!
Leave a Reply
Comments (0)