I've always been captivated by the stories of scientists who stood at the precipice of the unknown, peering into realms that challenged every established notion of reality. Among these titans, few embody the profound struggle and ultimate triumph of scientific discovery quite like Max Planck. His name is synonymous with the birth of quantum mechanics, a theory so revolutionary it fundamentally reshaped our understanding of the universe. Yet, what many don't realize is that Planck himself was a conservative physicist, deeply rooted in classical physics, who initially resisted the very radical ideas his own work unveiled. This blog post will be an extensive journey, a deep dive into the extraordinary life of Max Planck, from his early intellectual pursuits to the groundbreaking discoveries that earned him the Nobel Prize, and the immense personal tragedies he endured while witnessing the tumultuous 20th century unfold. Prepare for a very long read, as we unravel the complete tapestry of a life dedicated to truth, even when that truth was profoundly unsettling.
The Formative Years: A Classical Education (1858-1879)
Max Karl Ernst Ludwig Planck was born on April 23, 1858, in Kiel, Schleswig-Holstein, then part of Denmark. His family was steeped in a tradition of scholarship and public service. His father, Johann Julius Wilhelm Planck, was a professor of constitutional law at the University of Kiel, and his mother, Emma Patzig, came from a family of pastors and scholars. This intellectual heritage undoubtedly laid a strong foundation for young Max's future.
When Planck was nine years old, his family moved to Munich, where his father took up a professorship at the University of Munich. It was here that Max attended the Königliches Maximiliansgymnasium. I imagine him as a bright, diligent student, though perhaps not a dazzling prodigy in the way some scientific legends are often portrayed. His early education was rigorous, focusing heavily on classics, mathematics, and natural sciences.
One influential figure during his Gymnasium years was Hermann Müller, a mathematician who introduced Planck to the concept of the conservation of energy. This fundamental principle, that energy cannot be created or destroyed, only transformed, left a profound impression on him. It instilled in him a lifelong reverence for universal, immutable laws of nature – a belief that would ironically make his later quantum discoveries so difficult for him to accept wholeheartedly at first.
In 1874, at the age of 16, Planck enrolled at the University of Munich. Initially, he harbored thoughts of becoming a classical philologist or a musician – he was an accomplished pianist and organist, and music remained a lifelong passion. However, he eventually chose physics, reportedly after a conversation with Philipp von Jolly, a physics professor at Munich. Jolly famously advised Planck against pursuing physics, stating that "in this field, almost everything is already discovered, and all that remains is to fill a few gaps." This anecdote, often cited, is a testament to the prevailing belief in the late 19th century that classical physics had largely completed its grand mission. Planck, with characteristic humility and determination, responded that he didn't wish to discover new worlds, but only to understand the existing known ones. This desire for understanding, rather than outright revolution, would define his early career.
During his university studies, Planck was primarily interested in the theoretical aspects of physics. He studied under prominent physicists like Gustav Kirchhoff and Hermann von Helmholtz in Berlin, both giants in thermodynamics and electromagnetism. However, I sense he found their lectures somewhat uninspiring, often preferring to study their original papers. It was during this period that he delved deeply into Rudolf Clausius’s work on thermodynamics, particularly the concept of entropy. This work resonated deeply with Planck, aligning with his desire for universal, absolute laws. He found an almost aesthetic beauty in the second law of thermodynamics, which states that the entropy of an isolated system always increases over time. For him, this was a fundamental truth governing the universe.
The Early Academic Career and Focus on Thermodynamics (1879-1890)
Planck completed his doctoral dissertation in 1879 at the University of Munich, at the remarkably young age of 21. His thesis, "On the Second Law of Thermodynamics," was a continuation of his deep fascination with entropy. He argued for the universality of the second law, believing it to be an absolute principle of nature, not merely a statistical one. This early work already demonstrated his independent thinking and his commitment to fundamental principles.
After receiving his doctorate, Planck became a *Privatdozent* (unpaid lecturer) at Munich, a common path for aspiring academics in Germany. He struggled financially, even contemplating abandoning academia for a career in music or engineering. However, his perseverance paid off. In 1885, he was appointed associate professor of theoretical physics at the University of Kiel, his birthplace. This was a significant step, providing him with a stable income and a platform to pursue his research.
During his time in Kiel, Planck continued his extensive research into thermodynamics, particularly its applications to physical chemistry. He investigated various phenomena, including phase transitions, solutions, and electrochemistry, always striving to find the underlying thermodynamic principles. His work during this period, while not yet revolutionary, solidified his reputation as a meticulous and insightful theoretical physicist. He was deeply conservative in his scientific outlook, valuing continuity and classical rigor above all else. This conservative approach, ironically, would make his later, more radical discoveries all the more impactful, as they were arrived at through painstaking logical deduction rather than speculative leaps.
In 1889, Planck was called back to the University of Berlin, initially as an associate professor, and then in 1892, he succeeded Gustav Kirchhoff as a full professor of theoretical physics. Berlin was then one of the leading centers of scientific research in the world, and this appointment placed Planck at the heart of the intellectual vibrant European physics community. He began teaching, drawing students with his clear and systematic lectures, even though I suspect his reserved demeanor might have initially presented a barrier for some.
It was in Berlin that Planck's research began to shift, albeit subtly, towards the problems that would eventually lead him to quantum theory. He was still very much a classical physicist, deeply entrenched in the ideas of continuous energy and deterministic laws.
Black-Body Radiation: The Seeds of Revolution (1890-1900)
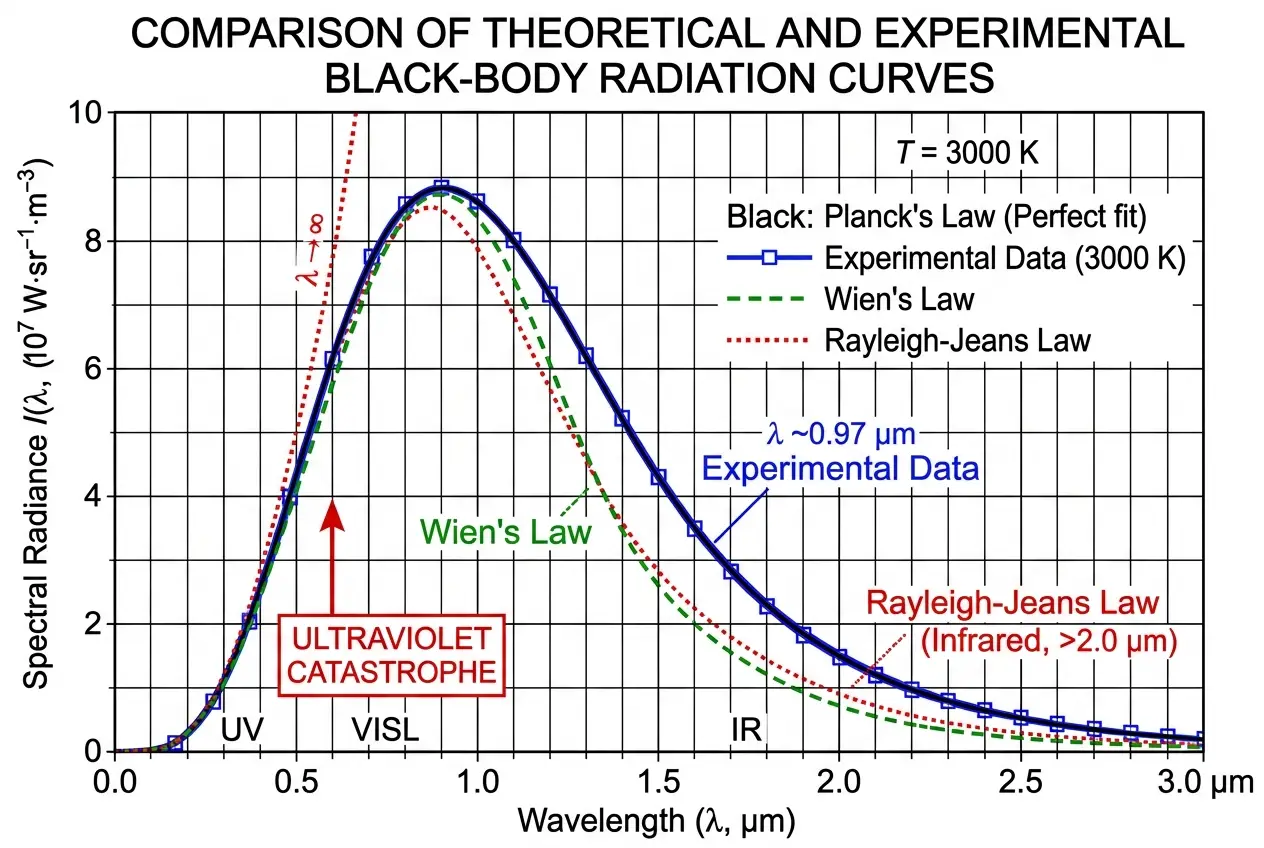
The turn of the 20th century was a fascinating time in physics. While many believed that the major puzzles had been solved, a few persistent anomalies resisted explanation by classical theories. One of the most perplexing of these was the problem of black-body radiation.
A "black body" is an idealized physical body that absorbs all incident electromagnetic radiation, regardless of frequency or angle of incidence. It also emits thermal radiation. Scientists were trying to understand the spectral distribution of this emitted radiation – that is, how much energy was radiated at different wavelengths – as a function of temperature. Experiments showed a specific curve, but classical physics couldn't fully explain it.
Two classical theories attempted to tackle this:
* **Wien's Law:** Worked well for high frequencies (short wavelengths) but failed at low frequencies.
* **Rayleigh-Jeans Law:** Worked well for low frequencies (long wavelengths) but predicted an infinite amount of energy at high frequencies, leading to the infamous "ultraviolet catastrophe." This catastrophic prediction highlighted a fundamental flaw in classical understanding.
I can only imagine the frustration of physicists grappling with this. It was an anomaly that screamed for a new approach. Planck, driven by his desire for absolute laws and a coherent understanding of nature, devoted himself to solving this problem. He was convinced there had to be a single, universal law that accurately described the entire black-body spectrum.
In 1900, after years of intense work, Planck introduced a new formula that perfectly matched the experimental data across all wavelengths. It was a remarkable achievement, but the derivation of this formula required a truly radical assumption. To make his formula work, Planck had to postulate that energy was not emitted or absorbed continuously, as classical physics assumed, but in discrete, indivisible packets, or "quanta." The energy (E) of each quantum was proportional to its frequency (ν), with the constant of proportionality being a new fundamental constant, which Planck famously denoted as *h*:
**E = hν**
This simple equation, now known as Planck's law, marked the birth of quantum mechanics. The constant *h*, now known as Planck's constant, is a tiny number (approximately 6.626 x 10
-34 Joule-seconds) that quantifies the relationship between energy and frequency at the quantum level.
Planck himself was deeply uneasy with this revolutionary idea. He viewed the quantization as a mathematical trick, a computational device, rather than a reflection of physical reality. He spent years trying to reconcile his quantum hypothesis with classical physics, hoping to find a way to make energy continuous again. He was a conservative by nature, and this break from classical thought was profoundly unsettling for him. As he later reflected, "I tried immediately to weld the elementary quantum of action somehow into the framework of classical theory, but it resisted all such attempts... I was quite alone in this attempt." His reluctance highlights the truly paradigm-shifting nature of his discovery. For more on the early ideas of quantum phenomena, one might explore the historical context of scientific thought on energy at
Wikipedia's page on Energy.
The Quantum Spreads: Reluctant Acceptance and Nobel Prize (1900-1918)
Despite Planck's initial reservations, his quantum hypothesis slowly began to gain traction, primarily because it worked so incredibly well. It was a young, audacious patent clerk named Albert Einstein who truly recognized the profound physical implications of Planck's work. In 1905, Einstein used Planck's quantum idea to explain the photoelectric effect, proposing that light itself consists of quanta (which he later called "photons"). This provided strong experimental evidence that quantization was not just a mathematical artifact but a fundamental aspect of nature. Einstein's work essentially turned Planck's uncomfortable hypothesis into a physical reality. You can read more about Einstein's pivotal contributions in our previous article,
Albert Einstein: The Mind Who Rewrote Reality.
Over the next decade, other physicists built upon Planck's and Einstein's insights. Niels Bohr used quantization to develop his model of the atom in 1913, successfully explaining the discrete spectral lines of hydrogen. The quantum revolution was underway, even if its reluctant father, Max Planck, still hoped it might somehow be reabsorbed into classical theory.
Planck's influence, even as he grappled with the implications of his own work, grew steadily. He became a respected elder statesman of German physics, serving as Rector of the University of Berlin from 1915 to 1916. He was also a passionate advocate for science and education.
In 1918, Max Planck was awarded the Nobel Prize in Physics "in recognition of the services he rendered to the advancement of Physics by his discovery of energy quanta." This award solidified his place in history, acknowledging the fundamental importance of his contribution, even as the full implications of quantum mechanics were still being explored and debated. It was a well-deserved honor for a man whose meticulous pursuit of truth, even when it led him to uncomfortable conclusions, had opened up an entirely new realm of physics.
| Year |
Key Event in Planck's Life |
Scientific Significance |
| 1858 |
Born in Kiel, Germany |
Beginning of a scientific journey |
| 1879 |
Doctorate from University of Munich |
Early work on Thermodynamics |
| 1889 |
Professor at University of Berlin |
Established as a leading theoretical physicist |
| 1900 |
Introduces Planck's Law and Quantum Hypothesis |
Birth of Quantum Mechanics |
| 1905 |
Einstein uses quanta to explain Photoelectric Effect |
Confirms physical reality of Planck's quanta |
| 1918 |
Awarded Nobel Prize in Physics |
Official recognition for discovery of energy quanta |
| 1930-1937 |
President of Kaiser Wilhelm Society |
Leadership role in German science during a tumultuous era |
| 1947 |
Died in Göttingen, Germany |
Leaves behind a monumental scientific legacy |
Personal Life and Tragedies (Early 20th Century)
While Planck's scientific achievements were soaring, his personal life was marked by immense tragedy. He married Marie Merck in 1887, and they had four children: Karl, Grete, Emma, and Erwin. His family was central to his life, and he cherished them deeply.
In 1909, his wife Marie died, leaving him with four young children. This was a profound blow. He remarried in 1911 to Marga von Hoesslin, his first wife's niece, and they had one son, Hermann.
The First World War brought unimaginable sorrow. His eldest son, Karl, was killed in action in 1916. Just two years later, in 1917, his daughter Grete died during childbirth, and tragically, her newborn child also did not survive. As if that wasn't enough, in 1919, his other daughter, Emma, also died during childbirth, and her child too perished. To lose three children and two grandchildren within such a short span must have been utterly devastating. I can hardly fathom the emotional resilience required to continue his intellectual work in the face of such profound personal loss. This period highlights the stark contrast between the intellectual rigor of his scientific pursuit and the brutal reality of human suffering.

Planck and the Rise of Nazism (1930s-1940s)
The 1930s brought a new, darker set of challenges to Germany and, by extension, to Planck. As the Nazis rose to power, their ideology began to infect all aspects of German society, including science. Planck, as the president of the Kaiser Wilhelm Society (now the Max Planck Society), the leading scientific institution in Germany, found himself in an impossible position.
He was a man of deep integrity and believed fiercely in the autonomy and international character of science. He tried, with limited success, to protect Jewish scientists who were being purged from their positions. He personally appealed to Adolf Hitler to reconsider the policy of dismissing Jewish scientists, arguing that these individuals were essential to Germany's scientific prowess. Hitler, famously, responded with an uncompromising rejection, stating that if dismissing Jewish scientists meant the end of German science, then they would do without it for a few years.
Planck maintained a difficult stance, trying to preserve German science while avoiding open confrontation that he believed would only worsen the situation for scientists. He advised some Jewish colleagues to emigrate, helping them secure positions abroad, while others, like Fritz Haber (a Nobel laureate whose work on nitrogen fixation was crucial for fertilizer production, but who was Jewish), faced public humiliation and exile. I imagine the immense moral dilemma he faced – how to protect the institution of science and its people without directly challenging a brutal, totalitarian regime. His efforts, though often insufficient in the face of Nazi ruthlessness, demonstrate his moral courage. The complex relationship between science and politics during this era is a subject explored in depth on
Wikipedia's article on Science and Technology in Nazi Germany.
Despite his efforts, the scientific community in Germany was irrevocably damaged. Many brilliant minds left, and those who remained faced immense pressure. Planck, in a desperate attempt to show some form of resistance, subtly used his position to protect research and uphold academic standards where he could. However, the Nazi regime eventually stripped him of his presidency of the Kaiser Wilhelm Society in 1937, effectively pushing him out of public life.
World War II and Final Years (1940s)
The Second World War brought yet more devastation to Planck's life. He and his second wife remained in Germany, witnessing the destruction of their homeland. Their home in Berlin was destroyed by Allied bombing in 1944, along with many of his priceless scientific notes and books.
But the most crushing blow was yet to come. His beloved son, Erwin, a highly respected government official, was arrested in 1944 for his involvement in the failed July 20 Plot to assassinate Hitler. Despite Planck's frantic efforts to save him, Erwin was executed in January 1945. This was an unspeakable tragedy, the loss of his last surviving child, a testament to the barbarity of the regime he had quietly resisted.
At 87 years old, having lost his first wife, four children, and his home, Planck was a broken man, yet his spirit of inquiry remained. After the war, he was revered as a symbol of integrity and the enduring spirit of German science. He helped in the rebuilding efforts of German scientific institutions, and the Kaiser Wilhelm Society was renamed the Max Planck Society in his honor in 1948, a year after his death.
Max Planck passed away on October 4, 1947, in Göttingen, at the age of 89. He lived through two world wars, the rise and fall of totalitarianism, and witnessed the complete overhaul of physics that he himself had inadvertently initiated. His legacy is not just in the revolutionary equation E=hν, but in his unwavering commitment to truth, his profound moral courage in the face of adversity, and his enduring belief in the power of human reason.
Planck's Legacy: The Enduring Impact of Quantum Theory
Max Planck's reluctance to fully embrace the quantum was a paradox that defined his scientific journey. He was a classicist who inadvertently ushered in the quantum age. His work provided the foundational cornerstone upon which a new edifice of physics was built. Without E=hν, there would be no modern understanding of atoms, molecules, light, or matter.
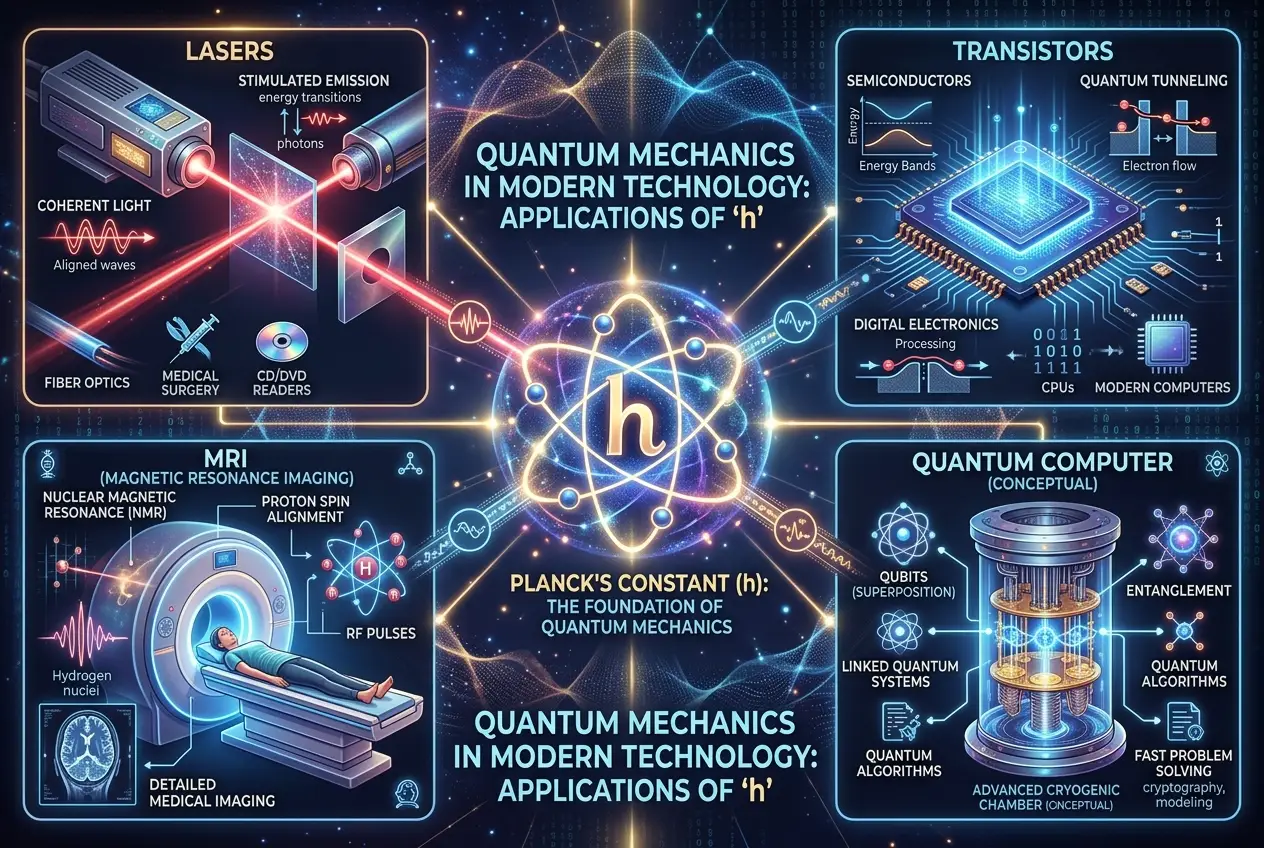
Quantum mechanics, the theory he founded, is now one of the most successful and rigorously tested theories in all of science. It underpins virtually all of modern technology:
* **Lasers:** From barcode scanners to fiber optics, lasers are a direct application of quantum principles.
* **Transistors:** The building blocks of all modern electronics, from smartphones to supercomputers, rely on quantum mechanics to function.
* **Medical Imaging:** Technologies like MRI (Magnetic Resonance Imaging) use quantum phenomena to peer inside the human body.
* **Quantum Computing:** A nascent field, quantum computing harnesses phenomena like superposition and entanglement to potentially solve problems currently intractable for classical computers. This area of research is particularly fascinating, and I recall a blog post we published earlier:
Black Holes: Are They Nature's Ultimate Quantum Computers?, which touches upon the profound computational implications of quantum phenomena. We also delved into
Is Empty Space a Quantum Computer?, further illustrating the vast reach of quantum theory.
Planck's constant, *h*, is now one of the most fundamental constants of nature, appearing in virtually every equation of quantum mechanics. It defines the scale at which quantum effects become significant. For instance, understanding how quantum sensors operate can reshape our reality perception, as explored in
Do Quantum Sensors Reshape Our Reality Perception?.
His legacy also extends beyond physics. He exemplified the ideal of a scientist as a seeker of truth, driven by an inherent curiosity and a rigorous methodology. His personal suffering and his moral stance during the Nazi era remind us that science is not conducted in a vacuum but is deeply intertwined with human values and societal challenges.
I believe his journey offers a powerful lesson: even the most conservative and rigorous minds can, through their unwavering commitment to objective truth, stumble upon realities that shatter their most cherished assumptions. Planck's reluctance to embrace the quantum initially wasn't a flaw; it was a testament to the revolutionary nature of his discovery and the profound intellectual courage it took to publish something so fundamentally at odds with the established scientific order. He opened a door to a universe stranger and more complex than anyone had imagined, a universe where energy comes in discrete packets, and reality is governed by probabilities. For a deeper understanding of the conceptual underpinnings of quantum mechanics, the
Wikipedia page on Quantum Mechanics provides an excellent resource.
In a world increasingly driven by rapid innovation, Planck's story reminds me that true breakthroughs often emerge from meticulous, sustained inquiry, sometimes leading us to uncomfortable truths that demand a re-evaluation of everything we thought we knew. His life was a testament to the power of intellectual honesty and the profound, sometimes tragic, journey of discovery.

 Join Us
Join Us





































Leave a Reply
Comments (0)