The tapestry of modern physics is woven with threads of extraordinary intellect, groundbreaking discoveries, and profound philosophical debates. Among these luminaries, one figure stands particularly tall, his legacy so foundational that it reshaped our understanding of reality itself: Niels Henrik David Bohr. I find it astonishing how one mind could so elegantly bridge the classical world with the bewildering realm of the quantum, charting a course for an entirely new scientific paradigm. This blog post will be an extensive journey through the remarkable life and monumental contributions of Niels Bohr, a narrative spanning his early intellectual awakenings to his enduring philosophical impact on science and society. We will delve into the intricate details of his journey, exploring the personal struggles, the scientific triumphs, and the deep friendships that defined his path.
### The Genesis of a Genius: Early Life and Education (1885-1911)
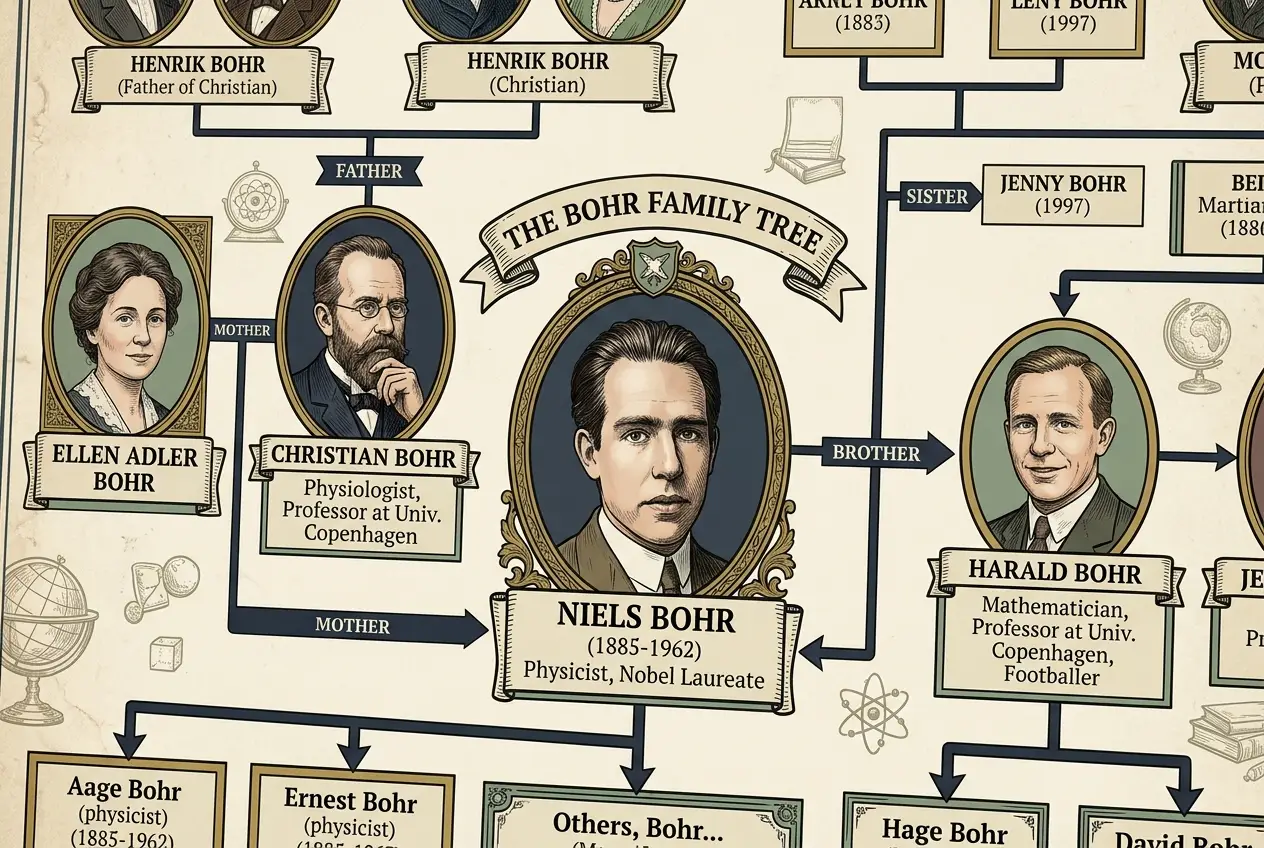
Niels Bohr was born on October 7, 1885, in Copenhagen, Denmark, into an environment steeped in intellectual pursuit and academic excellence. His father, Christian Bohr, was a distinguished professor of physiology at the University of Copenhagen, renowned for his work on respiration. His mother, Ellen Adler Bohr, hailed from a prominent Jewish family deeply involved in banking and parliamentary politics. This upbringing provided Niels and his younger brother, Harald (who would become a noted mathematician), with a stimulating intellectual playground, fostering lively discussions on science, philosophy, and culture from an early age. I often ponder how much of a child's environment shapes their future, and in Bohr's case, it seems pivotal.
The Bohr household was a hub of intellectual activity, frequently visited by prominent scientists and thinkers of the era. Christian Bohr's laboratory was practically an extension of their home, exposing young Niels to scientific inquiry and experimentation firsthand. I can only imagine the dinner table conversations, likely brimming with challenging questions and deep insights, shaping the minds of the future physicist and mathematician.
Niels attended the Gammelholm Latin School, where he excelled in mathematics and physics. His academic prowess was evident early on, though he was known for his thoughtful, sometimes slow, deliberation on problems rather than quick, flashy answers. This methodical approach would become a hallmark of his scientific method.
In 1903, Bohr enrolled at the University of Copenhagen, following in his father's footsteps but with a focus on physics. Here, he studied under Professor Christian Christiansen, a pioneer in experimental physics, and eventually under Professor Martin Knudsen. It was during this period that Bohr truly began to hone his skills, delving deep into the theoretical underpinnings of physics. His earliest significant work, while still a student, involved a theoretical and experimental investigation into the surface tension of water using oscillating fluid jets. This meticulous study, which earned him a gold medal from the Royal Danish Academy of Sciences and Letters in 1907, showcased his exceptional talent for both theoretical analysis and experimental design. His master's degree followed in 1909, and he completed his Doctor of Philosophy in 1911 with a dissertation titled "Studies on the Electron Theory of Metals." This thesis, a highly technical work, demonstrated his firm grasp of classical electromagnetic theory and hinted at his burgeoning discomfort with its limitations when applied to atomic phenomena. He was already sensing the cracks in the classical edifice that would soon become gaping chasms.
### Apprenticeship with Giants: Cambridge and Manchester (1911-1913)
Upon completing his PhD, Bohr realized that Denmark, despite its academic quality, was not yet at the forefront of the burgeoning field of atomic physics. The real action was in England, home to some of the most influential physicists of the time. In 1911, with a fellowship from the Carlsberg Foundation, Bohr embarked on what would become a pivotal scientific pilgrimage, first to Cambridge and then to Manchester.
His initial destination was the Cavendish Laboratory at the University of Cambridge, led by J.J. Thomson, the discoverer of the electron. Bohr was eager to collaborate with Thomson, particularly given his own doctoral work on the electron theory of metals. However, the collaboration did not unfold as smoothly as he might have hoped. Thomson, a towering figure in his own right, seemed less interested in Bohr's theoretical work, and communication proved challenging, partly due to Bohr's English language skills and his inherently deep, often complex, manner of expressing ideas. I can empathize with the frustration of a brilliant young mind feeling unheard by an established giant.
Despite the initial difficulties, Bohr's time at Cambridge was not without merit. He attended lectures, engaged with other researchers, and continued to grapple with the inconsistencies between classical physics and the emerging experimental data about atoms. It was during this period that he became aware of Ernest Rutherford's groundbreaking atomic model, often referred to as the "planetary model," where electrons orbited a tiny, dense, positively charged nucleus. Rutherford had proposed this model based on his famous gold foil experiment conducted in Manchester.
Sensing a better alignment with his interests, Bohr decided to move to Manchester in 1912 to work with Rutherford directly. This proved to be a transformative decision. Rutherford, known for his warm demeanor and openness to new ideas, was highly receptive to Bohr's inquiries. The atmosphere in Rutherford's laboratory was vibrant and conducive to exploration, a stark contrast to the more rigid environment Bohr experienced at Cambridge. Rutherford’s model, while revolutionary, had a fatal flaw from a classical perspective: according to classical electromagnetism, an orbiting electron should continuously radiate energy and spiral into the nucleus, rendering atoms unstable. The very existence of stable atoms, therefore, presented an insurmountable paradox for classical physics.
Bohr immediately recognized this central problem. During his time in Manchester, immersed in discussions with Rutherford and his colleagues, the seeds of his revolutionary atomic theory began to sprout. He started to conceptualize a radical departure from classical physics, seeking to incorporate Max Planck’s quantum hypothesis, which suggested that energy is emitted and absorbed in discrete packets or "quanta."
### The Quantum Leap: The Bohr Model of the Atom (1913)
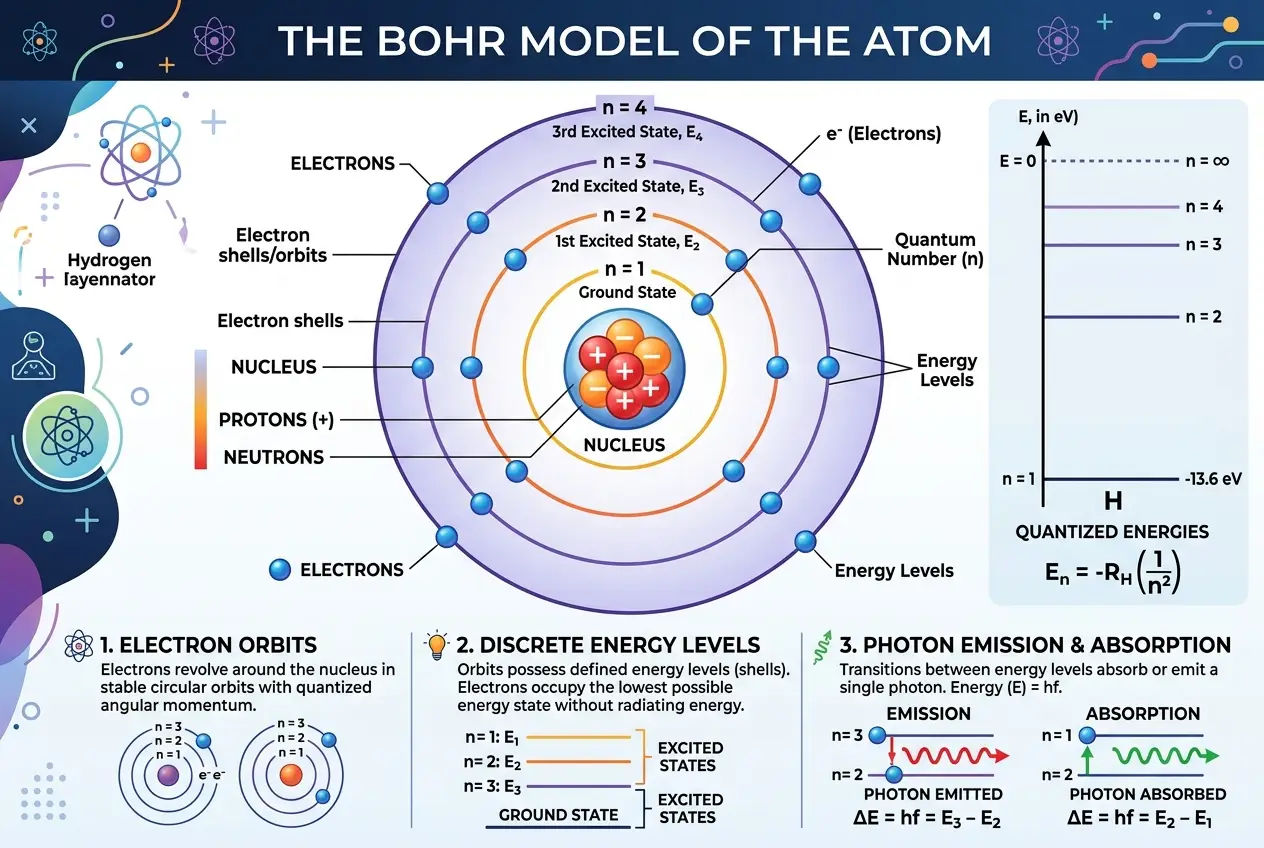
The year 1913 marked the publication of Bohr's seminal papers, a trilogy of articles in *Philosophical Magazine* that introduced what is now known as the Bohr Model of the Atom. These papers, "On the Constitution of Atoms and Molecules," fundamentally altered the course of physics. He proposed a model that was a brilliant, yet somewhat ad-hoc, fusion of classical mechanics and the nascent quantum theory.
The Bohr model was built upon several revolutionary postulates:
1. **Stationary States:** Electrons orbit the nucleus in specific, stable orbits (or "stationary states") without radiating energy. This directly contradicted classical electrodynamics. Each orbit corresponded to a fixed energy level.
2. **Quantized Angular Momentum:** The angular momentum of an electron in its orbit is quantized, meaning it can only take on certain discrete values, specifically integer multiples of Planck's constant divided by 2π (ħ). This was a direct application of Planck's ideas to atomic structure.
3. **Quantum Jumps (Transitions):** Electrons can jump from one stationary state to another by absorbing or emitting a photon of a specific energy. The energy of the photon is precisely equal to the energy difference between the two states (ΔE = hf, where h is Planck's constant and f is the frequency of the radiation).
4. **Correspondence Principle (later articulated):** For large orbits (or high quantum numbers), the quantum theory should merge with classical physics. This principle became a guiding light for Bohr in extending quantum ideas.
Using these postulates, Bohr was able to remarkably calculate the spectral lines of hydrogen with unprecedented accuracy. The discrete lines observed in the hydrogen spectrum had long been a mystery, and Bohr’s model provided a clear, quantitative explanation. This was a monumental triumph, giving physicists a working model that could explain atomic stability and spectral emissions, even if it defied classical intuition. I often imagine the sheer excitement and bewilderment among physicists as they first encountered these ideas.
The success of the Bohr model was immediate and profound. It provided a framework for understanding atomic structure and spectra that was previously lacking. It also introduced the concept of quantum numbers to describe the discrete energy levels of electrons. However, Bohr himself recognized the limitations of his model. It worked perfectly for hydrogen (a single-electron system) and hydrogen-like ions (e.g., He+, Li2+), but it struggled with multi-electron atoms and couldn't explain the varying intensities of spectral lines or the Zeeman effect (the splitting of spectral lines in a magnetic field) fully.
Despite its shortcomings, the Bohr model was a crucial stepping stone. It introduced the revolutionary idea that atoms exist in quantized energy states and that transitions between these states involve discrete packets of energy. It paved the way for the development of a more complete quantum mechanics. As a testament to its impact, Bohr was awarded the **Nobel Prize in Physics in 1922** "for his services in the investigation of the structure of atoms and of the radiation emanating from them." You can read more about his Nobel Prize and the historical context on [Wikipedia](https://en.wikipedia.org/wiki/Niels_Bohr).
### The Copenhagen Interpretation and the Quantum Debates (1920s)
After his groundbreaking work on the atomic model, Bohr returned to Copenhagen and, with the support of the Danish government and the Carlsberg Foundation, established the Institute for Theoretical Physics in 1921 (now known as the Niels Bohr Institute). This institute quickly became a Mecca for theoretical physicists, attracting a constellation of brilliant minds from around the world, including Werner Heisenberg, Wolfgang Pauli, Paul Dirac, and many others who would shape the future of quantum mechanics.
It was within the stimulating environment of the Copenhagen Institute that Bohr, along with Heisenberg and others, developed what came to be known as the **Copenhagen Interpretation of Quantum Mechanics**. This philosophical framework sought to make sense of the new, often counter-intuitive, mathematical formalism of quantum theory, particularly after Heisenberg formulated matrix mechanics in 1925 and Erwin Schrödinger developed wave mechanics in 1926.
The Copenhagen Interpretation is characterized by several key tenets:
1. **Wave-Particle Duality:** Quantum entities (like electrons, photons) exhibit both wave-like and particle-like properties, depending on how they are observed. They are neither purely one nor the other.
2. **Probability and Indeterminism:** Quantum mechanics does not provide deterministic predictions for individual events but only probabilities. The outcome of a quantum measurement is inherently probabilistic.
3. **Measurement Problem and Collapse of the Wave Function:** The act of measurement plays a crucial role. Before measurement, a quantum system exists in a superposition of states. The act of measurement "collapses" this superposition into a single, definite outcome.
4. **Complementarity Principle:** This was Bohr's most profound philosophical contribution. It states that certain properties of quantum systems, like position and momentum, or wave and particle nature, are complementary. One cannot observe both simultaneously with arbitrary precision; gaining information about one necessarily limits the information one can gain about the other. These are not contradictory but rather mutually exclusive aspects of a single underlying reality.
The Copenhagen Interpretation, particularly the principle of complementarity, became the dominant interpretation of quantum mechanics for decades. However, it was not universally accepted, leading to one of the most famous scientific debates in history: the **Bohr-Einstein Debates**.
Albert Einstein, a titan of classical physics and creator of relativity, was deeply troubled by the probabilistic nature of quantum mechanics and the seemingly central role of the observer. His famous quote, "God does not play dice with the universe," encapsulated his discomfort. He believed in an objective reality where physical properties existed independently of measurement and that quantum mechanics, while successful, was an incomplete theory. He spent years devising thought experiments (like the EPR paradox, co-authored with Boris Podolsky and Nathan Rosen) to expose perceived inconsistencies or hidden variables in quantum mechanics.
Bohr, with his profound understanding of quantum phenomena and his philosophical acumen, consistently defended the completeness of quantum mechanics and the necessity of the Copenhagen Interpretation. In their famous debates at the Solvay Conferences, Bohr systematically dismantled Einstein’s arguments, often showing how Einstein’s thought experiments inadvertently upheld the very principles he sought to challenge, particularly the uncertainty principle. These debates were not about proving one side wrong, but rather a deeply philosophical exploration of the nature of reality at its most fundamental level. For a deeper look into the philosophical implications, you can refer to the [Stanford Encyclopedia of Philosophy entry on the Copenhagen Interpretation](https://plato.stanford.edu/entries/qm-copenhagen/).
Bohr's ability to articulate the profound implications of quantum mechanics, even when they defied common sense, cemented his place as not just a brilliant physicist but also a leading philosophical thinker of the 20th century. I believe his contribution here was not just scientific, but truly existential, forcing humanity to rethink its place in the universe.
### The Golden Age of Copenhagen: Nurturing a Generation of Scientists
The Institute for Theoretical Physics in Copenhagen, under Bohr's leadership, became an unparalleled center of scientific collaboration and innovation. It was a unique environment where young, aspiring physicists from across the globe came to learn, discuss, and contribute to the rapidly evolving field of quantum mechanics. Bohr was not just a director; he was a mentor, a facilitator, and a constant source of inspiration.
His working style was highly collaborative. He believed firmly in the power of dialogue and discussion, often engaging in lengthy, intense conversations with his students and colleagues, walking through parks, or sketching ideas on blackboards. Many accounts describe Bohr's process as one of constant refinement, often rewriting papers multiple times, seeking the most precise and unambiguous expression of complex ideas. This meticulousness, while sometimes frustrating for his collaborators, ensured clarity and depth in their work.
The "Copenhagen Spirit" referred to the informal, open, and intensely creative atmosphere that permeated the Institute. Here, the brightest minds freely exchanged ideas, debated fiercely but respectfully, and pushed the boundaries of human knowledge. Key developments in quantum mechanics, such as Heisenberg's uncertainty principle, Pauli's exclusion principle, and Dirac's relativistic equation, were either born or significantly refined within this intellectual crucible. Many of these scientists, including Heisenberg, Pauli, and Dirac, would go on to win Nobel Prizes themselves, a testament to Bohr's profound influence as a mentor.
Bohr also extended a helping hand to many refugee scientists fleeing political turmoil in Europe during the 1930s. He actively assisted Jewish scientists escaping Nazi Germany, finding them positions and offering them sanctuary in Copenhagen or helping them emigrate to other countries. This humanitarian aspect of his leadership further cemented his reputation as a deeply compassionate and ethical scientist. His institute was not just a scientific powerhouse but also a beacon of hope and intellectual freedom in a darkening world. I find this aspect of his life incredibly moving; his commitment to human dignity was as profound as his commitment to scientific truth.
### Nuclear Fission and World War II (1930s-1945)
The 1930s brought new discoveries that would have profound and tragic implications for the world. In 1938, German chemists Otto Hahn and Fritz Strassmann discovered nuclear fission, the process by which a heavy atomic nucleus splits into lighter nuclei, releasing an enormous amount of energy. Lise Meitner and Otto Frisch provided the theoretical explanation for this phenomenon.
Niels Bohr was among the first physicists to fully grasp the significance of fission. In January 1939, he traveled to the United States to lecture on recent developments, and it was there, at a physics conference, that he shared the news of fission with American physicists. His colleague, John Wheeler, collaborated with him to develop the liquid drop model of the nucleus, which was crucial for understanding the mechanism of fission. They theorized that it was the rarer isotope, Uranium-235, rather than the more abundant Uranium-238, that was primarily responsible for slow neutron fission. This distinction was critical for the development of nuclear weapons.
As World War II escalated and the implications of fission became terrifyingly clear, Bohr found himself in an increasingly precarious position. Denmark was occupied by Nazi Germany in April 1940. Despite the dangers, Bohr chose to remain in Copenhagen, continuing his work and discreetly aiding persecuted scientists. His institute became a symbol of intellectual resistance against the Nazi regime.
However, as the Nazis intensified their persecution of Jews and political opponents, Bohr, whose mother was Jewish, became a target. In September 1943, he received a warning that he was about to be arrested. With the help of the Danish resistance, Bohr and his family made a dramatic escape across the Øresund Strait to Sweden in a fishing boat. From Sweden, he was flown to England in a specially adapted Mosquito bomber, enduring a perilous journey where he nearly suffocated due to an ill-fitting oxygen mask.
Upon arriving in England, Bohr was immediately drawn into the Allied efforts to develop an atomic bomb. He was given the pseudonym "Nicholas Baker" and eventually traveled to the United States to work on the Manhattan Project, though his role was primarily as a consultant and a moral voice. He worked at the Los Alamos Laboratory, though his primary contribution was more conceptual and philosophical rather than directly technical.
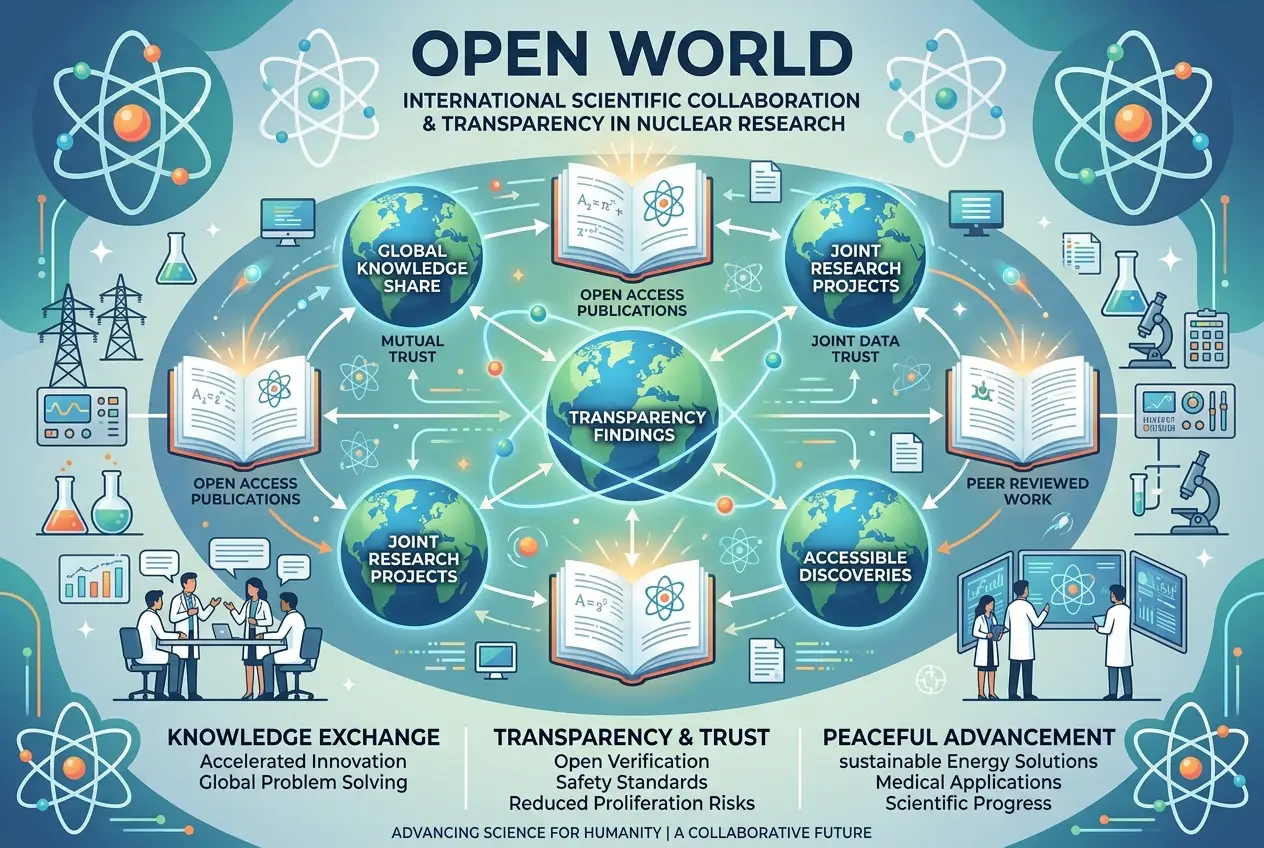
Bohr was deeply troubled by the prospect of nuclear weapons. He recognized the immense power they represented and the existential threat they posed to humanity. He advocated for **international cooperation and openness** regarding nuclear research, believing that only transparency could prevent a post-war arms race. He met with Winston Churchill and Franklin D. Roosevelt to press his case for an "Open World," arguing that the only way to control the atomic threat was for nations to share scientific knowledge and establish international oversight. His proposals, however, were met with skepticism and ultimately rejected by the Allied leaders, who prioritized military secrecy. This period truly highlights the moral quandaries faced by scientists when their discoveries have such profound societal implications. I can only imagine the burden on his conscience.
### Post-War Years and the "Open World" (1945-1962)
After the war, Bohr returned to Copenhagen and dedicated himself to promoting the peaceful uses of atomic energy and advocating for an "Open World" policy. He believed that the atomic bomb had fundamentally changed the nature of international relations, necessitating a new approach based on mutual trust and understanding rather than secrecy and competition. His 1950 open letter to the United Nations, a plea for the abolition of nuclear weapons and the promotion of international scientific cooperation, encapsulated this vision.
Though his immediate proposals for an "Open World" were not adopted by political leaders, Bohr remained a tireless advocate for disarmament and peace. He became involved in various international initiatives, including the establishment of CERN (the European Organization for Nuclear Research) in 1954, which was envisioned as a center for international scientific collaboration, a direct embodiment of Bohr's ideals. He also helped establish Nordita (Nordic Institute for Theoretical Physics) in 1957, further cementing Copenhagen's role as a leading center for physics.
Bohr continued his scientific work, delving deeper into the philosophical foundations of quantum mechanics, particularly the concept of complementarity, which he extended beyond physics into other fields of knowledge, including biology and psychology. He remained a respected and beloved figure in the scientific community, consistently hosting scientists and participating in conferences worldwide. His influence extended far beyond his direct scientific contributions, shaping the ethical and philosophical discourse surrounding science in the atomic age.
In 1957, he was awarded the first **Atoms for Peace Award**, a recognition of his efforts to harness nuclear energy for humanitarian purposes. This award underscored his lifelong commitment to using science for the betterment of humanity, despite his earlier involvement with the Manhattan Project.
### The Essence of Bohr's Philosophy: Complementarity
Bohr's concept of complementarity wasn't merely a solution to a paradox in quantum physics; it was a profound philosophical stance that he believed had implications far beyond the atomic realm. It suggested that certain seemingly contradictory descriptions are both necessary to fully understand a phenomenon, even if they cannot be observed simultaneously. For example, knowing an electron's precise position precludes knowing its precise momentum, and vice-versa. Neither description is more "real" than the other; both are aspects of a deeper, unified reality.
He extended this idea to other domains:
* **Biology:** He pondered how the mechanistic, reductionist view of life (understanding biological processes through physics and chemistry) could be complementary to a holistic, teleological view (understanding life in terms of purpose and function). Both are necessary to grasp the full complexity of living organisms.
* **Psychology:** He saw complementarity in the relationship between objective observation of brain processes and subjective experience of consciousness. We cannot fully observe our own brains in a mechanistic way at the same time we are having a conscious experience; they are complementary modes of understanding.
* **Culture and Society:** Bohr even suggested that complementarity could apply to different cultures or political ideologies, where seemingly opposing viewpoints might both contain valid truths, and a complete understanding requires appreciating both.
This pervasive philosophical perspective allowed Bohr to embrace the paradoxes of quantum mechanics rather than trying to force them into a classical mold. He argued that the language we use to describe phenomena is inherently limited by our classical experiences and that we must accept that at the quantum level, reality operates in ways that defy our macroscopic intuition. His thought challenged the very notion of a single, objective reality independent of observation, a radical departure that continues to spark debate among physicists and philosophers today. I find this aspect of his thinking truly captivating, as it elevates physics from a mere description of nature to a profound inquiry into the nature of knowledge itself.
### Personal Life and Character
Beyond his scientific achievements, Niels Bohr was known for his warm, jovial, and deeply humane personality. He married Margrethe Nørlund in 1912, and their marriage was a lifelong partnership marked by deep affection and mutual support. Margrethe played an invaluable role in his life, providing emotional stability and a practical counterbalance to his often abstract scientific thoughts. They had six sons, four of whom survived to adulthood. One of his sons, Aage Bohr, would also become a distinguished physicist and shared the Nobel Prize in Physics in 1975 for his work on the collective model of the atomic nucleus, making them one of the few father-son Nobel laureate pairs.
Bohr was a devoted family man, finding solace and joy in his home life. He was also an avid sportsman, particularly enjoying skiing and sailing. These activities provided him with a much-needed mental break from the intensity of his scientific work.
His colleagues often spoke of his extraordinary humility, his willingness to listen, and his genuine interest in others' ideas, regardless of their status. He was famous for his deep thinking, sometimes characterized by long pauses and careful articulation. He had a great sense of humor and enjoyed playful banter, which often helped to ease tension during intense scientific debates. His humanity shone through in every aspect of his life, from his mentorship of young scientists to his advocacy for peace.
Many anecdotes highlight his characteristic thoroughness. He would often rework a manuscript dozens of times, seeking the perfect expression. Werner Heisenberg famously recalled how Bohr's discussions could sometimes be exhausting but always ultimately clarifying, pushing one to the deepest understanding of a problem. His students and colleagues adored him, not just for his intellect but for his kindness and moral integrity.

### Awards and Legacy
Niels Bohr's contributions to physics were recognized with numerous honors throughout his life. As mentioned, he received the **Nobel Prize in Physics in 1922**. He was also elected a member of the Royal Society of London, the National Academy of Sciences (USA), and many other prestigious scientific academies around the world. He was awarded the Order of the Elephant, Denmark's highest order of chivalry.
His most enduring legacy, however, is not found in the accolades but in the fundamental concepts he introduced that continue to underpin modern physics:
* **The Quantized Atom:** His model, while superseded by more advanced quantum mechanics, established the idea of discrete energy levels and quantum jumps, providing the first successful explanation for atomic stability and spectra.
* **The Copenhagen Interpretation:** This framework profoundly shaped how physicists understood and talked about quantum mechanics, emphasizing wave-particle duality, probabilistic outcomes, and the role of measurement.
* **The Complementarity Principle:** A deep philosophical insight that continues to influence not just physics but also philosophy of science, encouraging a nuanced view of seemingly contradictory aspects of reality.
* **The Niels Bohr Institute:** A global center of excellence for theoretical physics, which continues to conduct cutting-edge research and foster international collaboration.
* **Mentor to a Generation:** His guidance and mentorship profoundly influenced many of the leading physicists of the 20th century, creating a lineage of quantum pioneers.
* **Moral Voice for Science:** His unwavering commitment to international cooperation and peaceful uses of atomic energy served as a powerful ethical compass for the scientific community in the nuclear age.
In many ways, Bohr helped humanity transition from a classical, deterministic view of the universe to a quantum, probabilistic one. He showed us that reality at its most fundamental level is stranger and more complex than we could have imagined, challenging our intuition and forcing us to develop new ways of thinking. His work laid the theoretical groundwork for countless technological advancements, from lasers and transistors to medical imaging and nuclear power.
### A Deeper Dive into the Bohr-Einstein Debates
To fully appreciate Bohr's intellectual depth, it's crucial to understand the intricate nuances of his debates with Albert Einstein. These were not personal squabbles but profound philosophical confrontations about the very nature of reality. Einstein, even after his revolutionary work on relativity and the photoelectric effect (which utilized Planck's quantum hypothesis), clung to the idea of a local, realistic, and deterministic universe. He believed in "elements of reality" that existed independently of observation. For more details on the historical context of these debates, see [Wikipedia's article on the Solvay Conference](https://en.wikipedia.org/wiki/Solvay_Conference).
Bohr, on the other hand, argued that at the quantum level, the classical concepts of reality simply break down. The act of measurement is not merely passive observation but an active interaction that helps define the reality being observed. His complementarity principle was his ultimate defense against Einstein's attempts to find "hidden variables" that would restore determinism.
Consider one famous thought experiment, the "photon box" proposed by Einstein at the 1930 Solvay Conference. Einstein imagined a box with a shutter that could open for a precisely controlled duration, allowing a single photon to escape. By weighing the box before and after, one could, in principle, determine the energy of the photon (via E=mc²), and by knowing the shutter's opening time, one could also determine the time of emission. This, Einstein argued, would allow simultaneous, precise measurement of energy and time, violating the energy-time uncertainty principle.
Bohr, after a sleepless night, found the flaw. He pointed out that to weigh the box accurately, one would need to introduce a clock into a gravitational field (if the weighing was done by suspending the box from a spring in a gravitational field). According to Einstein's own theory of general relativity, the rate of a clock depends on its position in a gravitational field. To measure the box's weight with arbitrary precision, one would have to precisely control its position, which, through general relativity's time dilation effects, would introduce an uncertainty in the measurement of time. Thus, Bohr used Einstein's own theory against him to show that the energy-time uncertainty principle held. This intellectual sparring showcases the extraordinary rigor and depth of both men.
**Table: Comparison of Classical and Quantum Perspectives (Bohr-Einstein Debates Context)**
| Feature | Classical Physics (Einstein's Preference) | Quantum Mechanics (Bohr's Interpretation) |
| :---------------- | :---------------------------------------------------- | :------------------------------------------------------------ |
| **Reality** | Objective, independent of observer | Dependent on context/measurement, fundamentally probabilistic |
| **Determinism** | Events are predetermined by initial conditions | Inherently probabilistic, outcomes are not fully predictable |
| **Causality** | Clear cause-and-effect chain | Challenges classical causality at quantum level |
| **Properties** | Intrinsic and well-defined (e.g., position, momentum) | Exist as potentialities until measured, complementary properties |
| **Measurement** | Passive observation, reveals pre-existing reality | Active interaction, influences/defines the measured reality |
| **Completeness** | QM is incomplete; hidden variables exist | QM is a complete description, no deeper reality to be found |
These debates highlighted a fundamental schism in how physicists viewed the very fabric of existence. While Einstein continued to seek a unified field theory that would incorporate quantum mechanics into a deterministic framework, Bohr remained steadfast in his conviction that quantum mechanics required a radical rethinking of our epistemological assumptions. The subsequent experimental verification of quantum entanglement, particularly through Bell's theorem, has largely sided with Bohr's view, indicating that local realism (Einstein's preference) is indeed violated at the quantum level. The implications of these debates continue to resonate in contemporary discussions about the foundations of quantum mechanics, including parallel universe theories and the nature of consciousness. You can explore a related topic on our own blog, about *decoding-reality-does-the-universe-hide-extra-dimensions-5269*.
### The Impact on Modern Technology
While Niels Bohr himself was primarily a theoretical physicist, his foundational work on atomic structure and quantum mechanics has had an immeasurable impact on modern technology. Every device that relies on understanding the behavior of electrons in atoms and molecules owes a debt to Bohr's early insights.
* **Semiconductors and Electronics:** The entire field of modern electronics, from microprocessors in our smartphones to the memory chips in our computers, relies on the quantum mechanical understanding of how electrons behave in solid materials. The band theory of solids, which explains conductivity, insulators, and semiconductors, is built upon the principles of quantized energy levels first introduced by Bohr.
* **Lasers:** Lasers operate on the principle of stimulated emission, where electrons jump between discrete energy levels in atoms or molecules, emitting photons of a specific wavelength. This precise control over light is a direct application of Bohr's atomic model. Lasers are ubiquitous, found in everything from barcode scanners and fiber-optic communication to surgical tools and data storage.
* **MRI (Magnetic Resonance Imaging):** This powerful medical diagnostic tool relies on the quantum mechanical properties of atomic nuclei, specifically their spin and how they interact with magnetic fields. The precise energy transitions of these nuclei, when excited by radio waves, allow for detailed imaging of soft tissues in the body.
* **Nuclear Energy and Weapons:** While a source of profound moral dilemma for Bohr, his early work on fission and the understanding of isotopes was crucial for both the development of nuclear weapons and, conversely, for the safe and controlled generation of nuclear power.
* **Materials Science:** The ability to design new materials with specific properties (e.g., superconductors, advanced alloys, nanomaterials) is deeply rooted in quantum mechanics, which allows scientists to predict and manipulate the interactions between atoms and electrons. The fascinating properties of materials like graphene, which we explored in *can-graphene-chips-unleash-ai-superpowers-8640*, are fundamentally quantum mechanical.
* **Quantum Computing:** The emerging field of quantum computing directly exploits exotic quantum phenomena like superposition and entanglement, whose theoretical foundations were laid by Bohr and his contemporaries. While still in its early stages, quantum computing promises to revolutionize computation, and its very existence traces back to the radical ideas born in Copenhagen. Our blog post *is-empty-space-a-quantum-computer-9021* further explores the mind-bending possibilities.
It is clear that Bohr's theoretical contributions were not abstract philosophical musings but fundamental insights that unlocked the secrets of the atom, paving the way for virtually all modern technological advancements that shape our daily lives.

### The Enduring Relevance of Bohr's Vision
Niels Bohr passed away on November 18, 1962, in Copenhagen, leaving behind a legacy that continues to inspire and challenge. His ideas didn't just provide answers; they taught us how to ask deeper, more fundamental questions about the universe and our place within it.
His commitment to open dialogue, international cooperation, and the ethical responsibility of scientists remains incredibly relevant in today's world, particularly with the rapid advancements in fields like AI and biotechnology. The questions he grappled with — about the nature of reality, the role of the observer, and the limits of human knowledge — are still at the forefront of scientific and philosophical inquiry.
The "Copenhagen Spirit" of collaborative inquiry and mutual respect serves as a timeless model for scientific progress. It reminds us that the greatest breakthroughs often emerge not from solitary genius, but from the dynamic interplay of diverse minds, openly debating and pushing against the boundaries of the known. I firmly believe that fostering such environments is crucial for solving the complex challenges humanity faces today.
Niels Bohr's life was a testament to the power of intellectual courage, human compassion, and unwavering curiosity. He was a pioneer who dared to venture into the unknown territory of the quantum, charting a course that transformed our understanding of the universe forever. His work compels us to embrace the wonder and the mystery, reminding us that reality, at its deepest levels, is often far more astonishing than our everyday perceptions suggest.
Verified Expert
Alex Rivers
A professional researcher since age twelve, I delve into mysteries and ignite curiosity by presenting an array of compelling possibilities. I will heighten your curiosity, but by the end, you will possess profound knowledge.
Leave a Reply
Comments (0)
No approved comments yet. Be the first to share your thoughts!
Leave a Reply
Comments (0)