I once watched a documentary about Earth's core, and it struck me how our planet itself is a colossal pressure cooker, relentlessly transforming matter into bizarre, extreme states. Imagine forces so immense they can reshape the very atoms of existence. This isn't just geological trivia; it's a frontier that could unlock a new generation of **super materials**, far beyond anything we use today.
We live in a world defined by the materials we can create. From the silicon in our smartphones to the composites in our spacecraft, progress often hinges on discovering or synthesizing substances with extraordinary properties. But what if the next leap isn't just about tweaking existing formulas, but about pushing the boundaries of physical reality itself? What if we could harness **extreme pressure** to forge materials with unimaginable strength, conductivity, or even entirely new functionalities, previously thought impossible?
The Unseen World of High Pressure Physics
When I think about pressure, my mind usually jumps to deep-sea submersibles or perhaps the incredible crushing force of a black hole. However, we're talking about something more controlled and systematic – the deliberate application of immense force to fundamentally alter the atomic structure of matter. This isn't a new concept, but the scale and precision we can achieve today are nothing short of revolutionary.
At Earth's surface, we experience roughly one atmosphere of pressure. But venture deep into the planet's mantle or core, and those numbers skyrocket. The center of Earth, for instance, endures pressures of over 3.6 million atmospheres (360 GPa) and temperatures reaching 6,000°C. Under these conditions, iron, nickel, and other elements behave in ways that would be utterly unrecognizable to us. They compress, their electron shells overlap, and they form new crystalline structures with vastly different properties. It's a natural laboratory of **material innovation**.
The Diamond Anvil Cell: Our Window into Extremes
So, how do scientists recreate these cosmic conditions in a lab? The answer often lies in a surprisingly elegant device: the **diamond anvil cell (DAC)**. This ingenious tool uses two perfectly aligned, gem-quality diamonds—the hardest known natural material—to squeeze tiny samples of matter. By applying force to the back of the diamonds, researchers can generate pressures exceeding those found at the Earth's core, sometimes reaching several terapascal (TPa) or tens of millions of atmospheres.
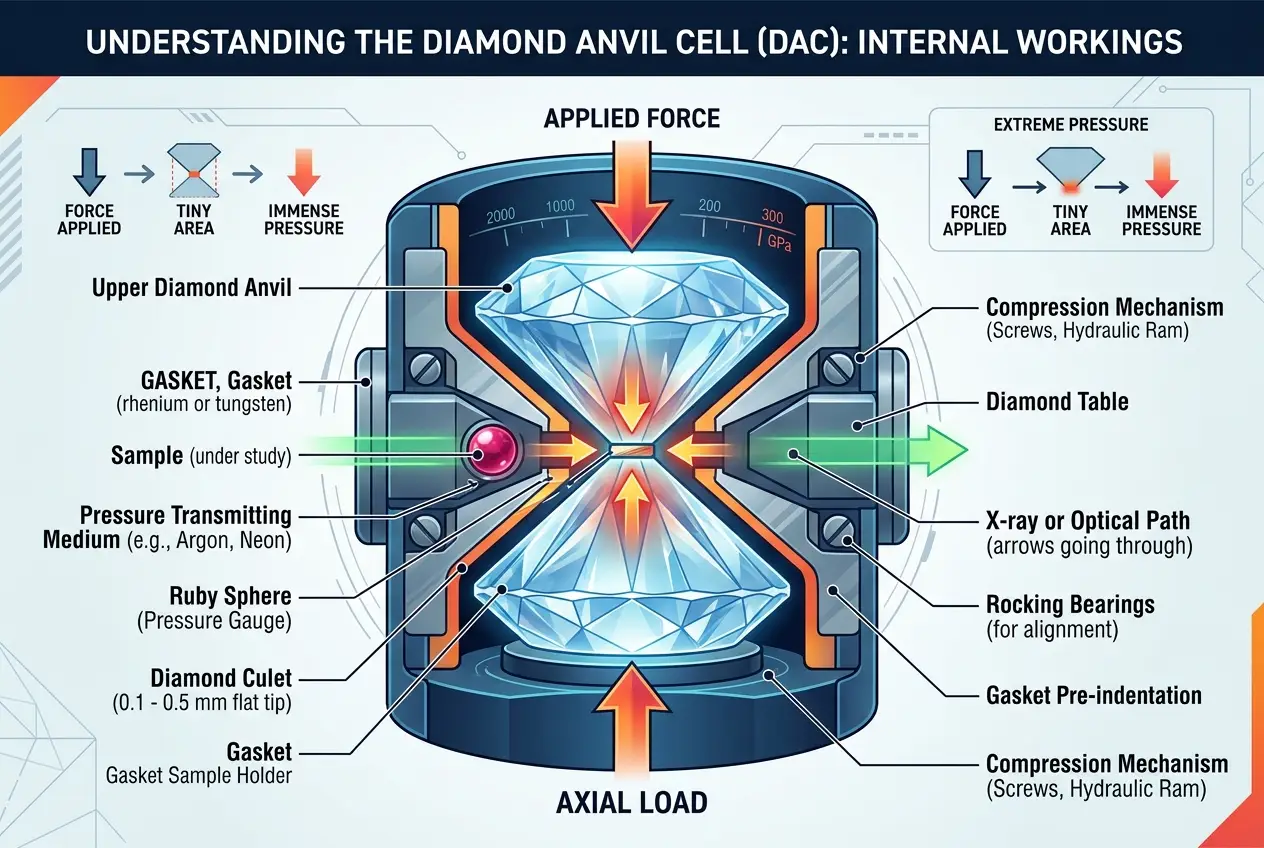
"The diamond anvil cell allows us to compress matter to densities and pressures comparable to those found deep inside planets, revealing entirely new states of matter," notes physicist Russell Hemley of George Washington University. This technology effectively allows us to peer into a realm where the ordinary rules of chemistry and physics bend and sometimes break. For more on this fascinating device, you can explore its capabilities on
Wikipedia's Diamond Anvil Cell page.
Beyond Diamonds: The Search for Exotic Phases
When we subject materials to such extreme pressures, several incredible things can happen:
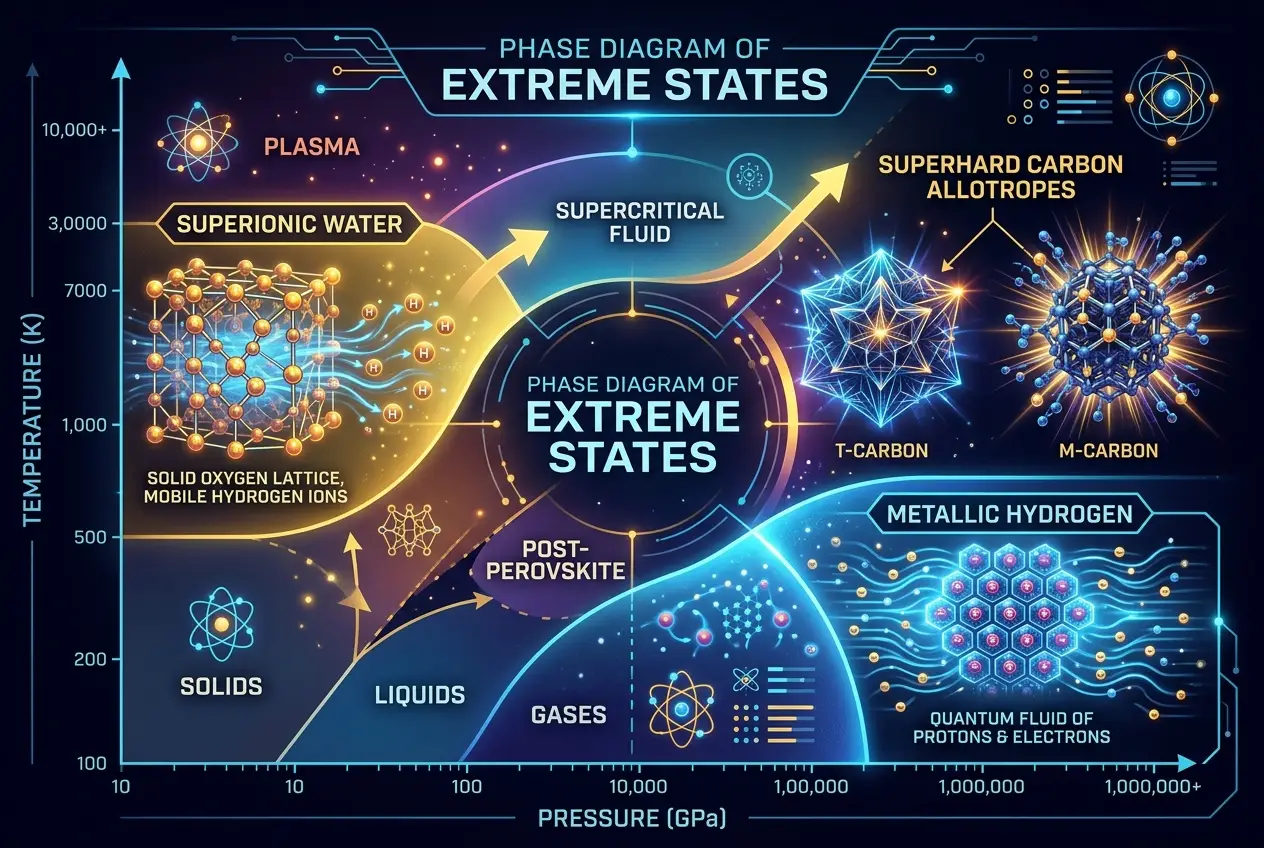
* **Phase Transitions:** A material can transform from one solid phase to another, or even become metallic when it wasn't before. For example, hydrogen, typically a gas, is predicted to become a superconducting metal at immense pressures.
* **New Compounds:** Atoms that wouldn't normally bond can be forced together to create entirely new chemical compounds with unique properties. Think about elements that are usually inert suddenly forming stable structures.
* **Altered Electronic Properties:** The spacing between atoms changes, altering how electrons move and interact. This can lead to materials that are superhard, superconductive at higher temperatures, or possess novel optical properties.
The Quest for Metallic Hydrogen
One of the most elusive and highly sought-after "supermaterials" is **metallic hydrogen**. Theoretically predicted over 80 years ago, this substance could be a superconductor at room temperature and pressure, an incredibly powerful rocket propellant, or even a pathway to understanding the interiors of gas giants like Jupiter. Imagine power lines with zero resistance, or spacecraft fueled by the most abundant element in the universe, condensed into an explosive solid.
While small samples of metallic hydrogen have been reportedly synthesized in DACs at pressures exceeding 495 GPa, maintaining its metallic state at lower pressures or temperatures remains a monumental challenge. The pursuit continues, pushing the boundaries of both experimental technique and theoretical understanding. This pursuit of exotic states of matter also links to discussions around
Metallic Hydrogen: Can it Unlock Unlimited Power?
Superconductivity and Superhard Materials
The discovery of high-temperature superconductors has been a holy grail in physics for decades. While current breakthroughs usually involve complex ceramics at cryogenic temperatures, extreme pressure offers another exciting route. By compressing certain materials, their atoms are forced closer, enhancing electron coupling and potentially leading to **superconductivity at much higher, even ambient, temperatures**. Imagine trains levitating effortlessly or lossless energy transmission.
Similarly, the quest for **superhard materials** extends beyond industrial cutting tools. Materials that can withstand extreme environments—be it in spacecraft, defense applications, or next-generation computing—are always in demand. Under immense pressure, known materials like carbon can transition into structures even harder than diamond, or entirely new, unyielding crystalline forms can emerge. For more insights into the potential of superhard materials, I recall a previous discussion on whether
Diamonds Hold Quantum Secrets.
Beyond Earth: Planetary Cores as Super Material Factories
Our own planet's interior gives us a glimpse of what's possible. But look further, to the cores of gas giants, rocky exoplanets, and even neutron stars, and the pressures become unfathomable. Jupiter's core, for instance, likely contains vast quantities of metallic hydrogen, and perhaps even 'diamond rain' in its atmosphere, as we discussed in
Diamond Rain: Can Planets Forge Cosmic Superconductors? On Neptune and Uranus, scientists speculate about superionic water, where oxygen atoms form a rigid lattice while hydrogen nuclei flow freely like a liquid metal. These are not just theoretical curiosities; they represent natural processes capable of forging materials far stranger and more robust than anything we can create today.
This naturally occurring extreme pressure on cosmic scales hints at the vast array of materials that could exist, challenging our conventional understanding of solids, liquids, and gases. Learning from these cosmic laboratories could inform our own high-pressure synthesis efforts.
The Future: Tailored Materials for Tomorrow's Challenges
The field of high-pressure material science isn't just about creating incredibly hard or conductive substances. It's about fundamentally reshaping our understanding of matter and opening doors to applications we can only begin to imagine.
**Think about:**
* **Next-generation electronics:** Imagine tiny components crafted from materials that conduct electricity with zero resistance, or optical elements that can manipulate light in unprecedented ways for quantum computing.
* **Energy storage and transmission:** Room-temperature superconductors could revolutionize power grids, making them infinitely more efficient and resilient.
* **Aerospace and defense:** Materials with unparalleled strength-to-weight ratios could lead to lighter, stronger spacecraft and impenetrable armor.
* **Medical advancements:** Biocompatible materials with unique properties could lead to revolutionary implants or diagnostic tools.
As I look at the incredible progress in this field, I can't help but feel a sense of wonder. We are, in essence, becoming cosmic alchemists, using immense pressure not to turn lead into gold, but to transform mundane elements into materials that defy expectation and unlock futuristic possibilities. The ability to precisely control these extreme conditions, coupled with advanced computational modeling, brings us closer than ever to designing and creating materials with properties tailored to our most ambitious technological challenges. This is where fundamental science meets transformative engineering, forging a future built on materials unseen just a few decades ago.
Conclusion
The journey into the high-pressure frontier is more than a scientific endeavor; it's a testament to human curiosity and our relentless drive to push the boundaries of knowledge. From the intricate workings of a diamond anvil cell to the crushing depths of planetary cores, the ability of extreme pressure to fundamentally alter matter holds the key to developing materials that were once confined to science fiction. As our understanding deepens and our techniques become more refined, I believe we stand on the cusp of forging a new era of technology, built literally from the atoms up, under conditions that mimic the very heart of stars.
&meta_title; Unseen Materials: How Extreme Pressure Forges Tomorrow's Tech &meta_title;
&meta_description; Discover the cutting-edge science of high-pressure physics and how it's forging new, unimaginable materials for future technology, from metallic hydrogen to superhard composites. &meta_description;
&faqs;{"faqs":[{"id":1,"question":"What exactly is \"extreme pressure\" in material science?","answer":"In material science, extreme pressure refers to conditions where materials are subjected to forces hundreds of thousands to millions of times greater than Earth's atmospheric pressure. These conditions are typically found deep inside planets or are recreated in laboratories using specialized equipment like diamond anvil cells."},{"id":2,"question":"How do scientists create such high pressures in a lab?","answer":"Scientists primarily use a device called a diamond anvil cell (DAC). It consists of two gem-quality diamonds with their tips (culets) facing each other. A tiny sample of material is placed between these tips, and mechanical force is applied to the back of the diamonds, concentrating immense pressure onto the sample."},{"id":3,"question":"What kind of new materials can be created under extreme pressure?","answer":"Extreme pressure can lead to the creation of materials with entirely new properties or phases. Examples include metallic hydrogen (a potential room-temperature superconductor), superhard forms of carbon even tougher than diamond, and exotic compounds from elements that normally don't bond. These materials often exhibit enhanced electrical conductivity, unique optical properties, or unprecedented strength."},{"id":4,"question":"What are the potential applications of these super materials?","answer":"The potential applications are vast. They include revolutionizing electronics (e.g., lossless power transmission with room-temperature superconductors), aerospace and defense (lighter, stronger materials for spacecraft and armor), energy storage (high-density fuels), and even medical advancements (new biocompatible materials)."},{"id":5,"question":"Is it possible for these materials, once formed, to remain stable at normal pressures?","answer":"The stability of these materials at normal pressures varies. Some materials undergo a reversible phase transition, meaning they revert to their original state once pressure is released. Others, like synthetic diamonds, can be quenched and remain stable at ambient conditions. The holy grail is to discover or synthesize materials that retain their extraordinary properties even after the extreme pressure is removed."}]}&faqs;

 Join Us
Join Us





































Leave a Reply
Comments (0)