The cosmos is a realm of unimaginable beauty and terrifying extremes. I often find myself lost in thought, picturing the swirling, turbulent atmospheres of gas giants, where pressures crush and temperatures scald. It's a place where matter behaves in ways we can barely replicate in our most advanced laboratories. Imagine, if you will, a storm so grand that it rains not water, but diamonds. Yes, actual diamonds. For years, scientists have theorized about this dazzling phenomenon on distant worlds, but what if these cosmic gem factories were doing more than just creating glittering carbon? What if they were forging materials with properties so extraordinary they could redefine our understanding of physics? This is the question that truly sparks my curiosity: **Could the diamond rain deep inside planets actually be creating natural superconductors?**
### The Cosmic Cauldron: Where Diamonds Fall Like Rain
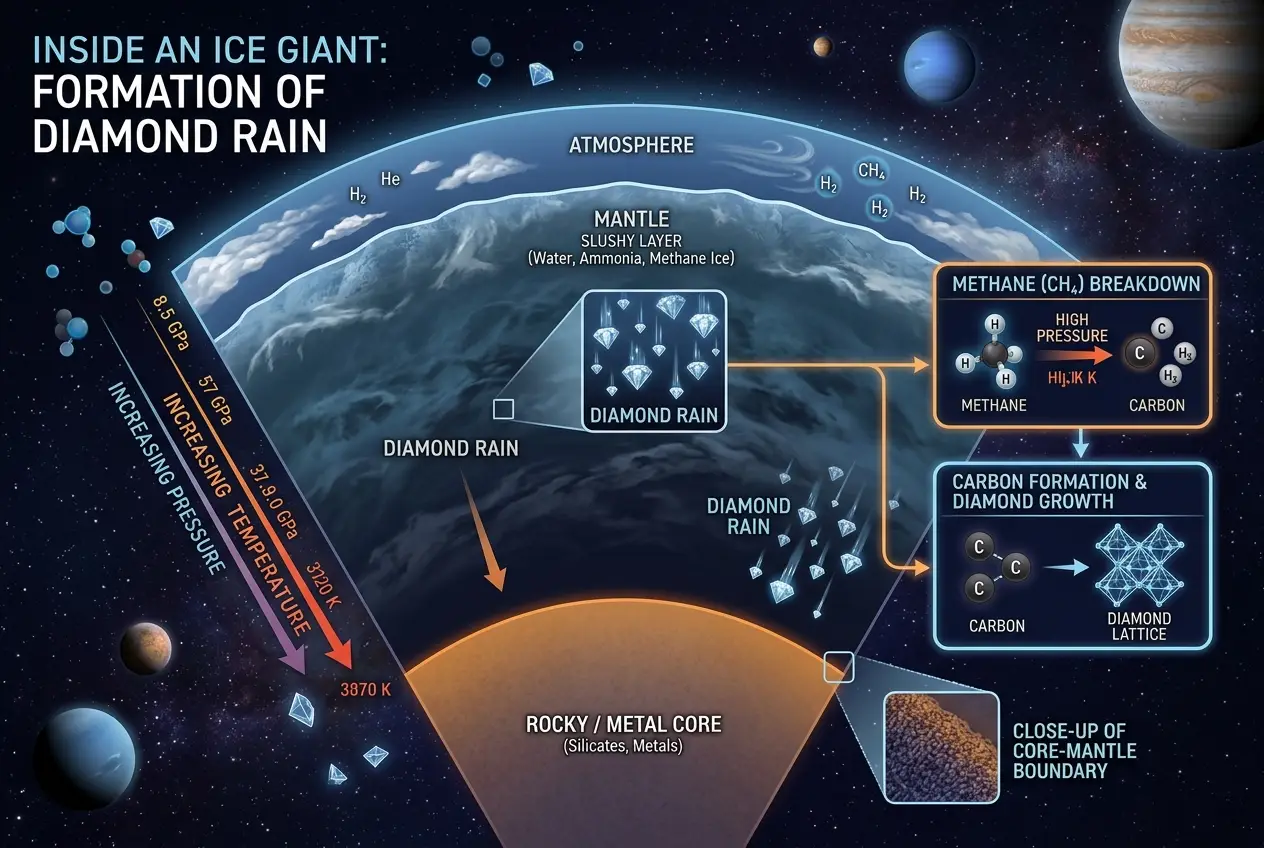
The concept of "diamond rain" isn't sci-fi fantasy; it's a compelling scientific hypothesis for the interiors of planets like Neptune and Uranus, and possibly even Jupiter and Saturn. These colossal worlds are not primarily rocky like Earth but are instead vast envelopes of hydrogen, helium, methane, and ammonia, all under incredible pressure.
Researchers first proposed the idea decades ago. They imagined the intense atmospheric pressures on ice giants like Neptune and Uranus – over a million times Earth's atmospheric pressure at sea level – compressing methane gas. As lightning storms, far grander than any on Earth, rip through these atmospheres, they could break apart methane (CH₄) molecules. This process would release carbon atoms. Under the crushing weight of the planet's layers, these free carbon atoms are then squeezed into crystalline structures, forming tiny, solid diamonds. And because of the immense gravitational pull, these diamonds would then literally "rain" down through the planet's mantle, possibly accumulating in vast liquid diamond oceans or even solid diamond cores.
Recent laboratory experiments have lent significant weight to this theory. Scientists have used powerful lasers to mimic the extreme pressures and temperatures found deep within these planets, successfully creating nanodiamonds from polystyrene, which simulates methane. This stunning breakthrough, detailed in articles like this one on [Wikipedia's Diamond Rain page](https://en.wikipedia.org/wiki/Diamond_rain), confirms that the conditions for diamond formation genuinely exist. It makes me wonder what other astonishing material transformations are happening out there, unseen.
### The Enigma of Superconductivity: A Glimpse into Zero Resistance
To understand why cosmic diamonds might hold a revolutionary secret, we first need to grasp the marvel that is superconductivity. A superconductor is a material that, when cooled below a certain critical temperature, can conduct electricity with **zero electrical resistance**. Imagine power lines that never lose energy, or electronics that generate no heat. This isn't just a minor improvement; it's a complete paradigm shift in how electricity behaves.
Beyond zero resistance, superconductors also exhibit perfect diamagnetism, a phenomenon known as the **Meissner Effect**, where they expel magnetic fields. This is what allows them to levitate above magnets. These properties are not just fascinating curiosities; they promise a future of ultra-efficient power grids, incredibly fast computing (think quantum computers), powerful MRI machines, and even magnetic levitation trains.
The catch? Most known superconductors require extremely low temperatures, often near absolute zero (-273.15 °C), achieved using expensive liquid helium or nitrogen. The quest for "room-temperature superconductors" (high-Tc superconductors) that work at more practical temperatures is one of the holy grails of material science. Such a discovery would fundamentally change our technological landscape. You can learn more about this groundbreaking field on [Wikipedia's Superconductivity page](https://en.wikipedia.org/wiki/Superconductivity). I often think about the grand challenges humanity faces in energy and technology; achieving room-temperature superconductivity would tackle many of them head-on.
### Extreme Physics: The Quest for Exotic States of Matter
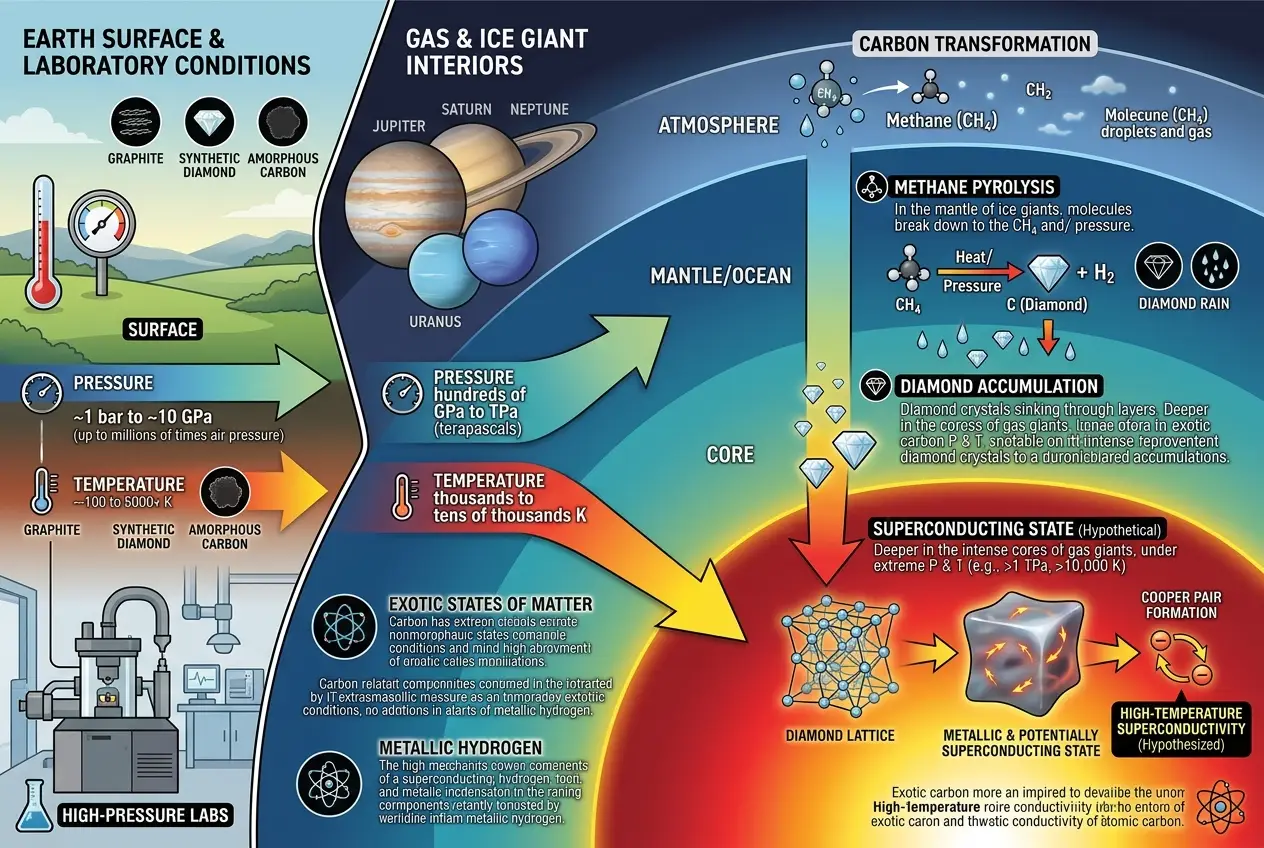
Planetary cores are the universe's ultimate pressure cookers. Under the incredible gravitational forces of gas and ice giants, ordinary matter is transformed into exotic states that defy our everyday experience. Hydrogen, for example, is theorized to become "metallic hydrogen" under the immense pressures found in Jupiter's core. In this state, hydrogen atoms lose their electrons, behaving like a metal and potentially becoming a superconductor at relatively high temperatures – perhaps even room temperature. This is a topic I’ve explored before, discussing similar extraordinary materials in the context of
Time Crystals and Exotic Matter.
The conditions for forming metallic hydrogen are similar to those for creating diamond rain, albeit at different depths and with different primary elements. The pressures are so intense that atomic bonds are rearranged, and electrons are forced into new configurations. This extreme environment is where the magic of potential cosmic superconductors truly lies. It's a realm where carbon, under pressure, forms diamonds, but what happens to those diamonds as they sink deeper, encountering even more unimaginable forces?
### A Hypothetical Forge: Could Diamond Cores be Superconducting?
Here's where the intrigue really deepens. If diamonds form and sink into regions of even higher pressure and temperature, could they undergo further structural changes that lead to superconductivity?
* **Pressure-Induced Superconductivity:** Scientists know that applying extreme pressure can induce superconductivity in materials that aren't typically superconductors. Hydrogen sulfide, for instance, has been shown to be a superconductor at record-high temperatures (around -70 °C) under extreme pressure. Could the incredible pressures deep within planetary cores, combined with the unique crystalline structure of diamonds, trigger similar or even more potent superconducting properties?
* **Carbon's Polymorphs:** Carbon is famously versatile, forming not just diamonds but also graphite, graphene, and fullerenes. Under extreme conditions, carbon can form even more exotic structures. Some theoretical models suggest that certain forms of carbon, when doped with other elements or under immense pressure, could exhibit superconductivity. If vast quantities of diamonds accumulate and are subjected to the core's unfathomable forces, perhaps a new, superconducting phase of carbon could emerge.
* **Interaction with Metallic Hydrogen:** In planets like Jupiter and Saturn, diamond formation might occur higher up, with a transition to metallic hydrogen deeper down. However, in the ice giants, the "diamond rain" might descend through layers of superionic water and ammonia, eventually mingling with a rocky core. The interaction between carbon and other elements in these super-pressurized, hot environments could create compounds that are ideal candidates for superconductivity. Such interactions between fundamental elements under extreme conditions offer a fascinating glimpse into material science at its most extreme.
While purely hypothetical at this stage, the possibility that nature's most dazzling gems might also be its most efficient conductors of electricity is a tantalizing thought. It reminds me of the grandeur of planetary phenomena, much like the mysteries of Jupiter's Great Red Spot, a colossal storm that has raged for centuries, still holding secrets about its longevity and dynamics, as discussed in our blog on
Jupiter's Great Red Spot.
### From Planetary Cores to Earthly Labs: The Pursuit of Room-Temperature Superconductors
The quest to understand planetary interiors isn't just about cosmic curiosity; it directly informs our search for advanced materials on Earth. If planets can spontaneously generate superconducting diamond structures or other exotic superconducting materials, it offers invaluable clues for laboratory synthesis.
Scientists are actively trying to recreate these extreme planetary conditions in diamond anvil cells, hoping to discover and synthesize new high-temperature superconducting materials. The insights gained from studying "diamond rain" and the behavior of carbon under mega-pressures could guide experiments, helping us unlock the secrets of room-temperature superconductivity. This global pursuit of a technological holy grail is one of humanity's most ambitious scientific endeavors, much like the ongoing mission to explore potential life on
Europa's hidden ocean.
Understanding how elements behave under such immense stress could revolutionize not just material science, but also our understanding of planetary evolution and even potential applications for future energy systems, perhaps even hinting at novel ways to generate power similar to discussions around
Antimatter as a future energy source.
### The Implications: Why Cosmic Superconductors Matter
If diamond rain truly does forge cosmic superconductors, the implications would be profound:
* **New Planetary Science:** It would reshape our models of planetary interiors, explaining magnetic fields, heat transfer, and evolutionary pathways in a new light. We might discover that planets are far more electrically active than we ever imagined.
* **Unlocking Material Secrets:** It would confirm that certain forms of carbon, or carbon compounds, can achieve superconductivity under specific conditions, providing a blueprint for creating such materials on Earth.
* **Inspiration for Future Technology:** The existence of naturally occurring room-temperature superconductors would ignite a new era of research and innovation, accelerating our progress towards energy efficiency, quantum computing, and advanced transportation systems. It challenges us to push the boundaries of materials engineering, much like the theoretical concepts behind
building a Dyson Sphere to harness a star's power.
The universe, in its boundless creativity, might already be demonstrating the ultimate in energy efficiency and material science, deep within the fiery hearts of distant worlds.
The idea that worlds beyond our own could be natural laboratories, silently churning out materials that we desperately seek here on Earth, is incredibly humbling and inspiring. The next time you look up at the night sky, consider the cosmic dance of elements, pressures, and temperatures happening billions of miles away. It might just be forging the superconductors of the universe, waiting for us to uncover their secret.

 Join Us
Join Us




































Leave a Reply
Comments (0)