I was recently engrossed in a documentary about astrobiology when a thought struck me: why do we always assume alien life, if it exists, must be like us? Our entire understanding of biology is built upon carbon, water, and a specific temperature range. But what if life, in its boundless cosmic variations, doesn't adhere to our terrestrial rulebook? What if, somewhere out there, entire ecosystems thrive not on organic compounds but on the very stuff of rocks – **silicon**? It’s a fascinating deviation from the norm, pushing the boundaries of what we consider "life" and challenging our deep-seated "carbon chauvinism."
The idea of silicon-based life isn't new. It’s been a staple of science fiction for decades, conjuring images of crystalline beings or sentient geological formations. But is there any scientific plausibility to it? Can silicon, a seemingly inert element, truly form the complex molecules necessary for metabolism, reproduction, and evolution? I find exploring these "what if" scenarios incredibly stimulating, as they force us to confront our biases and expand our imagination.
### The Foundation of Terrestrial Life: Carbon's Reign
To understand why silicon-based life is such a radical concept, we first need to appreciate carbon's unique role on Earth. Carbon is often called the **"king of chemistry"** for good reason. It has an unparalleled ability to form four stable covalent bonds with other atoms, including itself, creating long, diverse chains and rings. This versatility allows for an almost infinite array of complex organic molecules – from the sugars that fuel our cells to the DNA that carries our genetic code. Water, too, plays a crucial role as a universal solvent, facilitating chemical reactions and transporting nutrients within these carbon-based systems. Our entire existence, from the simplest bacterium to the most complex human, hinges on this elegant carbon-water partnership. For a deeper dive into organic chemistry, you might find this Wikipedia article on
Organic Chemistry insightful.
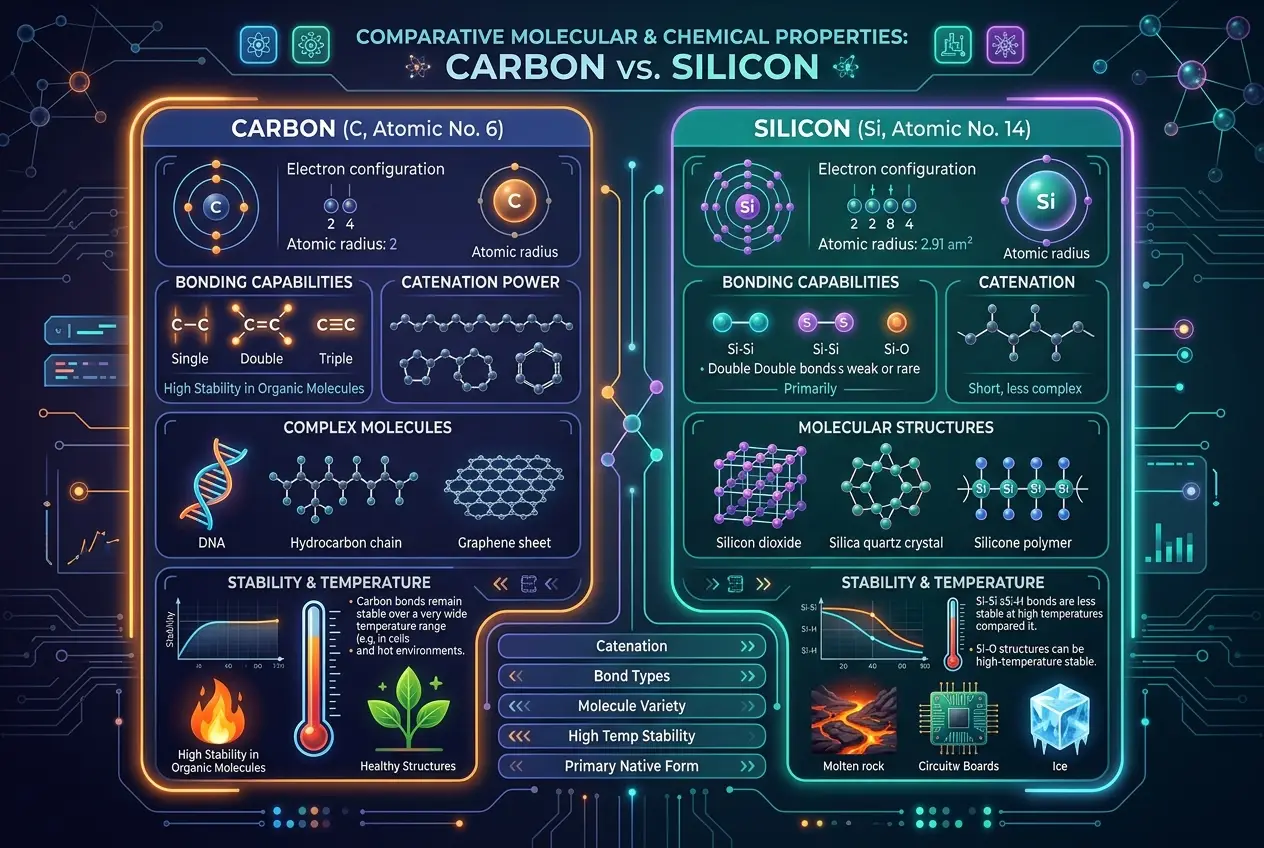
### Silicon: Carbon's Heavier Cousin
Now, let's turn to silicon. Located directly below carbon in the periodic table, silicon shares some superficial similarities. It can also form four bonds, and it's incredibly abundant, making up about 28% of Earth's crust. But that's where the easy comparisons end.
The crucial difference lies in silicon's atomic structure. Being a larger atom, its bonds with other elements are generally weaker and longer than carbon's. While carbon can readily form double and triple bonds, and complex long chains with itself (like hydrocarbons), silicon struggles to do the same. Silicon-silicon bonds are less stable, particularly in the presence of oxygen, where they prefer to form strong silicon-oxygen bonds, leading to the formation of silicates – the primary components of rocks and minerals. This is why our planet's crust is largely silicate-based, while life is carbon-based. The fundamental chemical properties diverge significantly, making it much harder for silicon to create the kind of complex, dynamic molecular machinery we see in carbon life.
### The Challenges of Silicon Biochemistry
Imagine trying to build a complex biological system using silicon. The hurdles are significant:
1. **Bond Stability:** Silicon's tendency to form strong bonds with oxygen makes it difficult for it to create the diverse, stable molecular backbones that carbon forms. Silicon-oxygen bonds are so stable that they tend to form rigid, unreactive solids like quartz, rather than flexible, dynamic biological structures.
2. **Solvent Issues:** If silicon is to replace carbon, it would likely need a different solvent than water. Water reacts strongly with many silicon compounds, breaking them down into silicates. Possible alternative solvents for exotic biochemistries include liquid ammonia, methane, or even sulfuric acid. Each of these presents its own challenges, often requiring extremely cold or hot environments.
3. **Energy Metabolism:** How would a silicon organism derive energy? Carbon-based life utilizes redox reactions, burning organic compounds for fuel. Silicon, however, doesn't easily participate in these reversible redox reactions that are fundamental to energy transfer in living systems. Could it rely on entirely different mechanisms, perhaps geothermal energy, or reactions with metallic compounds?
4. **Chirality:** Carbon's ability to form chiral molecules (molecules that are non-superimposable mirror images of each other, like our hands) is fundamental to life. Proteins, for example, are made exclusively of L-amino acids. Silicon's heavier nature might make it less adept at forming and distinguishing between chiral structures, which could complicate the assembly of complex biological machinery.
Despite these challenges, scientists continue to explore the theoretical possibilities. What if silicon's limitations in one area are compensated by advantages in another, especially under extremely different environmental conditions?
### Where Silicon Might Shine: Extreme Environments
The most compelling arguments for silicon-based life often place it in environments radically different from Earth's.
On planets with extremely high temperatures, where water would be a gas and organic molecules would decompose, silicon compounds might become more stable and reactive. For instance, molten salt or sulfuric acid oceans, or even incredibly dense, hot atmospheres, could serve as alternative solvents. In such conditions, silicon-oxygen bonds, which are problematic here, might become the very backbone of life, leading to organisms with crystalline or ceramic-like structures. Imagine life existing within molten rock or thriving near volcanic vents on an alien world.
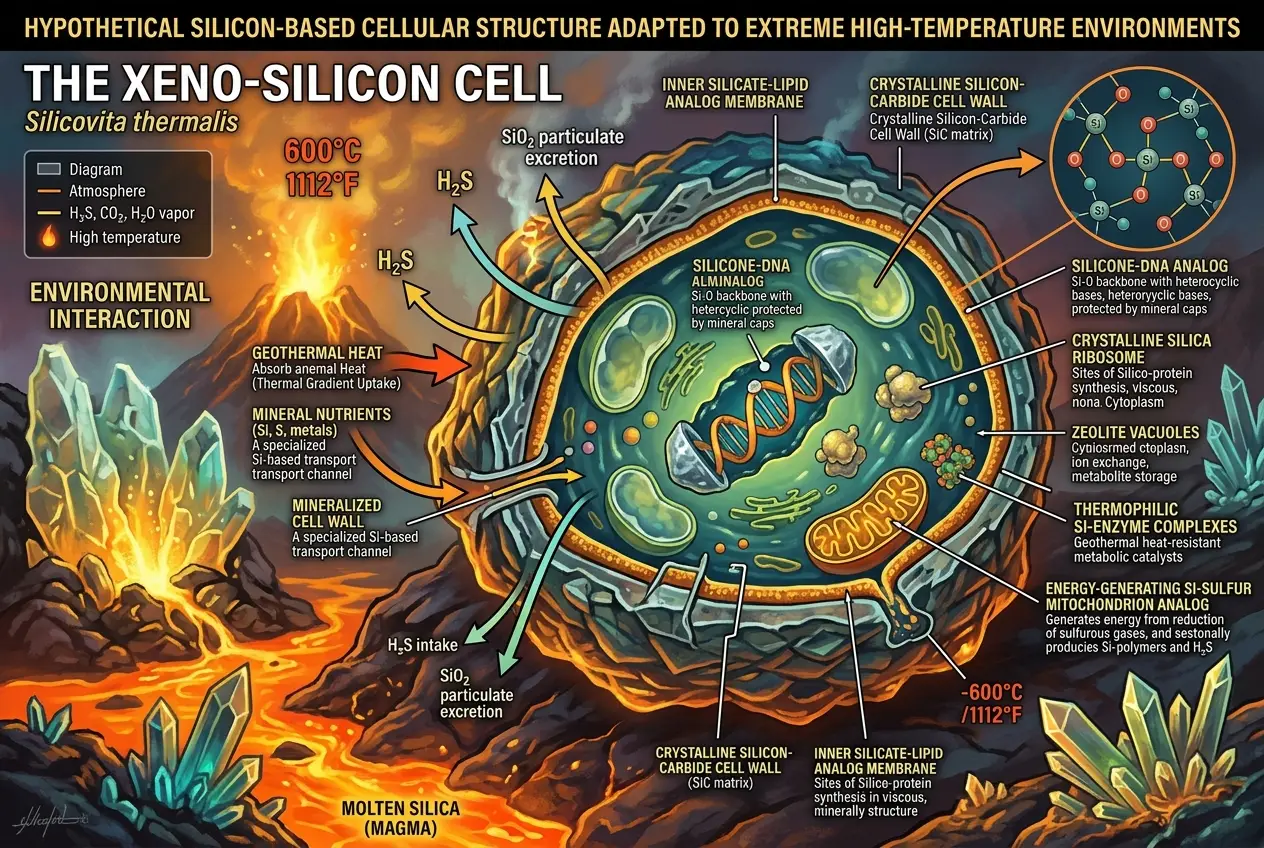
Some researchers have even suggested that silicon-based life might evolve on planets with very low temperatures, where liquid methane or ethane could act as solvents. In these frigid worlds, different chemical reactions might become viable. The key is that the conditions must be *so* different that carbon's advantages diminish, and silicon's alternative chemistry becomes feasible. For further reading on alternative biochemistries, I recommend checking out the Wikipedia entry on
Hypothetical Types of Biochemistry.
### The Search for the "Impossible" Organism
While pure silicon-based life remains theoretical, there are intriguing real-world examples that hint at its potential. Certain diatoms on Earth use silicon dioxide to construct their intricate cell walls, demonstrating life's ability to incorporate this element structurally. More recently, scientists have even engineered bacteria to create silicon-carbon bonds, a feat previously thought impossible for biological systems. This fascinating research, published in journals like *Science*, shows that life's chemical repertoire might be more flexible than we previously imagined. You can find more details on this groundbreaking work by searching for "biologically synthesized silicon-carbon bonds" or checking the
Wikipedia page on Silicon in biology. This pushes the boundaries of how we think about life's fundamental building blocks and potentially paves the way for synthetic silicon-containing organic molecules.
This area of research, often called **xenobiology** or **synthetic biology**, is a burgeoning field. While we haven't found a truly alien, silicon-based organism yet, the drive to understand what *could* be out there is pushing us to redefine our understanding of life itself. Could we one day encounter beings that metabolize minerals or communicate through crystalline lattice vibrations? It's a tantalizing prospect that reminds us how much more there is to discover beyond our Earthly confines.
### The Broader Implications for Astrobiology
The contemplation of silicon life dramatically expands the search parameters for alien civilizations. If life isn't confined to carbon and water, then the "habitable zone" around stars could be much larger and more diverse than we currently imagine. Planets previously deemed too hot, too cold, or too chemically volatile for life might suddenly become prime candidates for astrobiological exploration.
This perspective also encourages us to design more versatile life-detection technologies. Instead of solely searching for carbon signatures or water ice, future missions might need to look for a broader range of chemical and structural anomalies. What if some advanced civilizations are not "biological" in our sense, but have evolved into crystalline or geological entities, leveraging the stability and longevity of silicon? Perhaps we've been looking for organisms, when we should have been looking for an intelligent, self-organizing rock.
This quest to understand life's ultimate forms isn't just about discovering aliens; it's about understanding the very essence of existence. By questioning our assumptions about carbon, we open up a universe of possibilities, reminding us that reality is often far stranger and more magnificent than we dare to imagine. Just as ancient cultures pondered cosmic algorithms (a topic explored in previous articles like
Did Ancient Astronomers Decode Cosmic Time Algorithms?), we today are decoding the algorithms of life itself, in all its potential forms. And speaking of unusual forms of communication, the idea of life forms communicating in entirely non-biological ways brings to mind questions of digital anomalies, which you can read about in
Digital Anomalies: Glimpses of a Hidden Reality.
Could we ever truly identify silicon-based life? It would likely require us to look past our anthropocentric biases and develop instruments capable of detecting entirely new chemical signatures and metabolic processes. The discovery would be nothing short of revolutionary, proving that life is a cosmic phenomenon, adaptable and resilient beyond our wildest carbon-centric dreams.

In conclusion, while carbon remains the undisputed champion of life on Earth, the universe is vast, and its chemical possibilities are immense. The theoretical existence of silicon-based life pushes us to consider a broader definition of biology, challenging our preconceived notions and fueling our quest to understand life's true diversity across the cosmos. It’s a concept that truly sparks curiosity and wonder, prompting us to ask: what other incredible forms might life take?

 Join Us
Join Us






































Leave a Reply
Comments (0)